Folic acid - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for folic acid and what is the scope of freedom to operate?
Folic acid
is the generic ingredient in three branded drugs marketed by Ben Venue, Fresenius Kabi Usa, Xgen Pharms, Wyeth Pharms Inc, Amneal Pharm, Athem, Barr, Cadila Pharms Ltd, Chartwell Molecular, Contract Pharmacal, Everylife, Halsey, Hikma Pharms, Impax Labs, Ivax Sub Teva Pharms, Jubilant Cadista, Lannett, Leading, Lilly, Mk Labs, Nexgen Pharma Inc, Nuvo Pharms Inc, Pharmeral, Pioneer Pharms, Purepac Pharm, Qingdao Baheal Pharm, Sandoz, Sun Pharm Industries, Tablicaps, UDL, Usl Pharma, Valeant Pharm Intl, Vangard, Vintage, Vintage Pharms, Watson Labs, Whiteworth Town Plsn, and Mission Pharma, and is included in forty NDAs. Additional information is available in the individual branded drug profile pages.There are ten drug master file entries for folic acid. Twenty-seven suppliers are listed for this compound.
Summary for folic acid
| US Patents: | 0 |
| Tradenames: | 3 |
| Applicants: | 38 |
| NDAs: | 40 |
| Drug Master File Entries: | 10 |
| Finished Product Suppliers / Packagers: | 27 |
| Raw Ingredient (Bulk) Api Vendors: | 141 |
| Patent Applications: | 3,922 |
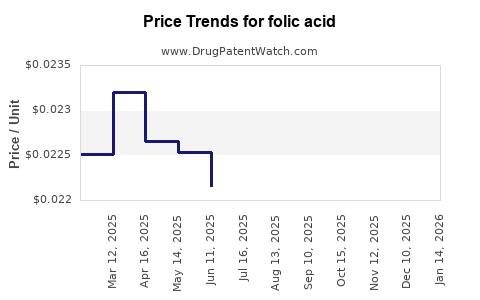
| Drug Prices: | Drug price trends for folic acid |
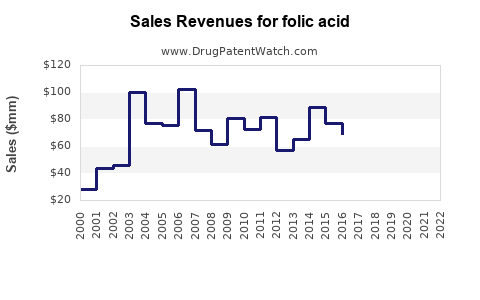
| Drug Sales Revenues: | Drug sales revenues for folic acid |
| What excipients (inactive ingredients) are in folic acid? | folic acid excipients list |
| DailyMed Link: | folic acid at DailyMed |
Medical Subject Heading (MeSH) Categories for folic acid
US Patents and Regulatory Information for folic acid
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sun Pharm Industries | FOLIC ACID | folic acid | TABLET;ORAL | 040582-001 | Jul 18, 2005 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mission Pharma | FOLICET | folic acid | TABLET;ORAL | 087438-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Wyeth Pharms Inc | FOLVITE | folic acid | TABLET;ORAL | 005897-004 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ben Venue | FOLIC ACID | folic acid | INJECTABLE;INJECTION | 081066-001 | Dec 29, 1993 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Halsey | FOLIC ACID | folic acid | TABLET;ORAL | 083598-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mk Labs | FOLIC ACID | folic acid | TABLET;ORAL | 083526-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |