Ucb Inc Company Profile
✉ Email this page to a colleague
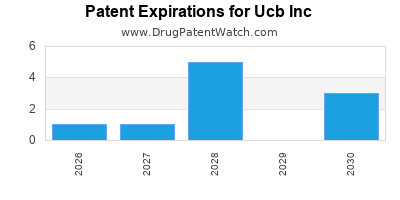
What is the competitive landscape for UCB INC, and when can generic versions of UCB INC drugs launch?
UCB INC has forty-two approved drugs.
There are thirty-four US patents protecting UCB INC drugs.
There are four hundred and eighty-four patent family members on UCB INC drugs in fifty-one countries and thirty-nine supplementary protection certificates in fourteen countries.
Summary for Ucb Inc
| International Patents: | 484 |
| US Patents: | 34 |
| Tradenames: | 29 |
| Ingredients: | 24 |
| NDAs: | 42 |
| Drug Master File Entries: | 1 |
| Patent Litigation for Ucb Inc: | See patent lawsuits for Ucb Inc |
Drugs and US Patents for Ucb Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ucb Inc | FINTEPLA | fenfluramine hydrochloride | SOLUTION;ORAL | 212102-001 | Jun 25, 2020 | RX | Yes | Yes | 10,947,183*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Ucb Inc | VIMPAT | lacosamide | SOLUTION;INTRAVENOUS | 022254-001 | Oct 28, 2008 | AP | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ucb Inc | FINTEPLA | fenfluramine hydrochloride | SOLUTION;ORAL | 212102-001 | Jun 25, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Ucb Inc | ZILBRYSQ | zilucoplan sodium | SOLUTION;SUBCUTANEOUS | 216834-002 | Oct 17, 2023 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-003 | May 12, 2016 | RX | Yes | No | 6,911,461 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Ucb Inc | VIMPAT | lacosamide | TABLET;ORAL | 022253-001 | Oct 28, 2008 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Ucb Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ucb Inc | KEPPRA | levetiracetam | TABLET;ORAL | 021035-003 | Nov 30, 1999 | 4,837,223 | ⤷ Try a Trial |
| Ucb Inc | UNIVASC | moexipril hydrochloride | TABLET;ORAL | 020312-001 | Apr 19, 1995 | 4,743,450 | ⤷ Try a Trial |
| Ucb Inc | VIMPAT | lacosamide | SOLUTION;INTRAVENOUS | 022254-001 | Oct 28, 2008 | 5,654,301 | ⤷ Try a Trial |
| Ucb Inc | NEUPRO | rotigotine | FILM, EXTENDED RELEASE;TRANSDERMAL | 021829-004 | Apr 2, 2012 | 6,884,434 | ⤷ Try a Trial |
| Ucb Inc | VIMPAT | lacosamide | SOLUTION;ORAL | 022255-001 | Apr 20, 2010 | 5,654,301 | ⤷ Try a Trial |
| Ucb Inc | VIMPAT | lacosamide | TABLET;ORAL | 022253-004 | Oct 28, 2008 | 5,654,301 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for UCB INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 10 mg/mL, 20 mL | ➤ Subscribe | 2016-06-30 |
| ➤ Subscribe | Orally Disintegrating Tablets | 0.25 mg, 0.5 mg, 1 mg and 2 mg | ➤ Subscribe | 2005-12-27 |
| ➤ Subscribe | Tablets | 1000 mg | ➤ Subscribe | 2007-01-24 |
| ➤ Subscribe | Tablets | 50 mg, 100 mg, 150 mg, and 200 mg | ➤ Subscribe | 2012-10-29 |
| ➤ Subscribe | Extended-release Tablets | 1000 mg | ➤ Subscribe | 2011-01-07 |
| ➤ Subscribe | Tablets | 7.5mg/12.5mg 15 mg/25 mg and 15 mg/12.5 mg | ➤ Subscribe | 2004-01-15 |
| ➤ Subscribe | Extended-release Transdermal Film | 1 mg/24 hr, 2 mg/24 hr, 3 mg/24 hr,4 mg/24 hr,6 mg/24 hr, and 8 mg/24 hr | ➤ Subscribe | 2013-11-26 |
| ➤ Subscribe | Oral Solution | 10 mg/mL | ➤ Subscribe | 2012-10-29 |
International Patents for Ucb Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Iceland | 6481 | ⤷ Try a Trial |
| Cyprus | 1124298 | ⤷ Try a Trial |
| Serbia | 60134 | ⤷ Try a Trial |
| Israel | 165918 | ⤷ Try a Trial |
| South Korea | 20190057420 | ⤷ Try a Trial |
| China | 1740150 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Ucb Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1265862 | PA2016013,C1265862 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: BRIVARACETAMAS; REGISTRATION NO/DATE: EU/1/15/1073 20160114 |
| 0162036 | 2000C/032 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LEVETIRACETAM; NAT. REGISTRATION NO/DATE: EU/1/00/146/001 20000929; FIRST REGISTRATION: CH 55297 20000329 |
| 1033978 | 24/2006 | Austria | ⤷ Try a Trial | PRODUCT NAME: ROTIGOTINE; REGISTRATION NO/DATE: EU/1/05/331/001 - EU/1/05/331/013 20060215 |
| 1429780 | 13C0012 | France | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE CIPROFLOXACINE ET DE DEXAMETHASONE, EN PARTICULIER DE CHLORHYDRATE DE CIPROFLOXACINE ET DE DEXAMETHASONE; NAT. REGISTRATION NO/DATE: NL 41308 20121214; FIRST REGISTRATION: 48976 20120808 |
| 0888289 | C300376 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LACOSAMIDE; REGISTRATION NO/DATE: EU/1/08/470/001-016 20080829 |
| 0888289 | SPC/GB09/007 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: LACOSAMIDE AND ITS PHARMACEUTICALLY ACCEPTABLE FORMS; REGISTERED: UK EU/1/08/470/001 20080829; UK EU/1/08/470/002 20080829; UK EU/1/08/470/003 20080829; UK EU/1/08/470/004 20080829; UK EU/1/08/470/005 20080829; UK EU/1/08/470/006 20080829; UK EU/1/08/470/014 20080829; UK EU/1/08/470/015 20080829; UK EU/1/08/470/016 20080829; UK EU/1/08/470/007 20080829; UK EU/1/08/470/008 20080829; UK EU/1/08/470/009 20080829; UK EU/1/08/470/010 20080829; UK EU/1/08/470/012 20080829; UK EU/1/08/470/013 20080829 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.