Sanofi Aventis Us Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SANOFI AVENTIS US, and what generic alternatives to SANOFI AVENTIS US drugs are available?
SANOFI AVENTIS US has one hundred and twenty-two approved drugs.
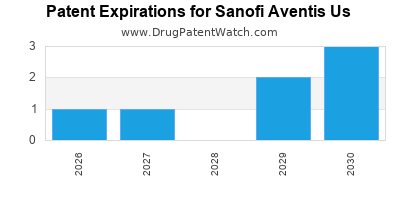
There are ten US patents protecting SANOFI AVENTIS US drugs.
There are two hundred and seventeen patent family members on SANOFI AVENTIS US drugs in fifty-two countries and one hundred and fifteen supplementary protection certificates in nineteen countries.
Summary for Sanofi Aventis Us
| International Patents: | 217 |
| US Patents: | 10 |
| Tradenames: | 97 |
| Ingredients: | 85 |
| NDAs: | 122 |
Drugs and US Patents for Sanofi Aventis Us
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sanofi Aventis Us | PENETREX | enoxacin | TABLET;ORAL | 019616-004 | Dec 31, 1991 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Sanofi Aventis Us | PARATHAR | teriparatide acetate | INJECTABLE;INJECTION | 019498-001 | Dec 23, 1987 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Sanofi Aventis Us | AMARYL | glimepiride | TABLET;ORAL | 020496-003 | Nov 30, 1995 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Sanofi Aventis Us | ISUPREL | isoproterenol hydrochloride | AEROSOL, METERED;INHALATION | 011178-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Sanofi Aventis Us | LASIX | furosemide | INJECTABLE;INJECTION | 016363-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Sanofi Aventis Us
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Sanofi Aventis Us | BRICANYL | terbutaline sulfate | TABLET;ORAL | 017618-002 | Approved Prior to Jan 1, 1982 | 3,937,838 | ⤷ Try a Trial |
| Sanofi Aventis Us | TALWIN NX | naloxone hydrochloride; pentazocine hydrochloride | TABLET;ORAL | 018733-001 | Dec 16, 1982 | 4,105,659 | ⤷ Try a Trial |
| Sanofi Aventis Us | PLAVIX | clopidogrel bisulfate | TABLET;ORAL | 020839-001 | Nov 17, 1997 | 4,847,265*PED | ⤷ Try a Trial |
| Sanofi Aventis Us | TAXOTERE | docetaxel | INJECTABLE;INJECTION | 020449-003 | Aug 3, 2010 | 4,814,470*PED | ⤷ Try a Trial |
| Sanofi Aventis Us | LOVENOX | enoxaparin sodium | INJECTABLE;INTRAVENOUS, SUBCUTANEOUS | 020164-009 | Jan 23, 2003 | RE38743 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for SANOFI AVENTIS US drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 7 mg and 14 mg | ➤ Subscribe | 2016-09-12 |
| ➤ Subscribe | Tablets | 300 mg/25 mg | ➤ Subscribe | 2006-06-06 |
| ➤ Subscribe | Tablets | 400 mg | ➤ Subscribe | 2013-07-01 |
| ➤ Subscribe | Injection | 5 mg/mL, 10 mL and 20 mL vials | ➤ Subscribe | 2007-02-09 |
| ➤ Subscribe | Injection | 200 mg/40 mL | ➤ Subscribe | 2007-07-16 |
| ➤ Subscribe | Injection | 100 mg/mL, 3 mL vials | ➤ Subscribe | 2006-12-07 |
| ➤ Subscribe | Extended-release Tablets | 12.5 mg | ➤ Subscribe | 2006-01-19 |
| ➤ Subscribe | Tablets | 150 mg/12.5 mg and 300 mg/12.5 mg | ➤ Subscribe | 2004-11-10 |
| ➤ Subscribe | Tablets | 75 mg, 150 mg and 300 mg | ➤ Subscribe | 2004-05-25 |
| ➤ Subscribe | Tablets | 300 mg | ➤ Subscribe | 2009-03-04 |
| ➤ Subscribe | Injection | 40 mg/mL, 0.5 mL and 2 mL vials | ➤ Subscribe | 2009-06-30 |
| ➤ Subscribe | For Injection | 50 mg/vial and 100 mg/vial | ➤ Subscribe | 2007-02-09 |
| ➤ Subscribe | Injection | 5 mg/mL, 40 mL vial | ➤ Subscribe | 2011-03-23 |
| ➤ Subscribe | Extended-release Tablets | 6.25 mg | ➤ Subscribe | 2006-02-24 |
International Patents for Sanofi Aventis Us Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Tunisia | SN06086 | ⤷ Try a Trial |
| New Zealand | 588623 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2011051894 | ⤷ Try a Trial |
| Australia | 2015200149 | ⤷ Try a Trial |
| Taiwan | I533866 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Sanofi Aventis Us Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0454511 | 10/1999 | Austria | ⤷ Try a Trial | PRODUCT NAME: ''IRBESARTAN'' ZUSAMMEN MIT ''HYDROCHLOROTHIAZID''; REGISTRATION NO/DATE: EU/1/98/086/001 - EU/1/98/086/006 19981015 |
| 3300601 | 2022C/528 | Belgium | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN DAUNORUBICINE EN CYTARABINE; AUTHORISATION NUMBER AND DATE: EU/1/18/1308 20180827 |
| 0281459 | 9890035-0 98910359 | Sweden | ⤷ Try a Trial | PRODUCT NAME: PLAVIX-CLOPIDOGREL; REG. NO/DATE: EU/1/98/069/001 19980715 |
| 0454511 | SPC/GB99/008 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: 2-N-BUTYL-4-SPIROCYCLOPENTANE-1-((2'-(TETRAZOL-5-YL)BIPHENYL-4-YL)METHYL)-2-IMIDAZOLIN-5-ONE)(GENERIC NAME IRBESARTAN) OPTIONALLY IN THE FORM OF ONE OF ITS SALTS AND HYDROCHLOROTHIAZIDE; REGISTERED: UK EU/1/98/086/001 19981015; UK EU/1/98/086/002 19981015; UK EU/1/98/086/003 19981015; UK EU/1/98/086/004 19981015; UK EU/1/98/086/005 19981015; UK EU/1/98/086/006 19981015 |
| 0281459 | 9890035 | Sweden | ⤷ Try a Trial | PRODUCT NAME: PLAVIX-CLOPIDOGREL; REG. NO/DATE: EU/1/98/069/001 19980715 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.