Pf Prism Cv Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PF PRISM CV, and what generic alternatives to PF PRISM CV drugs are available?
PF PRISM CV has sixteen approved drugs.
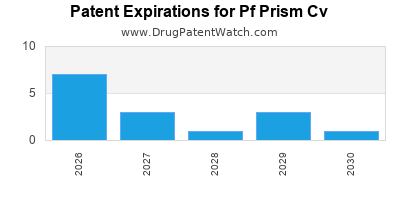
There are twenty-nine US patents protecting PF PRISM CV drugs.
There are six hundred and twelve patent family members on PF PRISM CV drugs in sixty-six countries and seventy-two supplementary protection certificates in eighteen countries.
Summary for Pf Prism Cv
| International Patents: | 612 |
| US Patents: | 29 |
| Tradenames: | 11 |
| Ingredients: | 11 |
| NDAs: | 16 |
Drugs and US Patents for Pf Prism Cv
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pf Prism Cv | BOSULIF | bosutinib monohydrate | CAPSULE;ORAL | 217729-002 | Sep 26, 2023 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pf Prism Cv | XALKORI | crizotinib | CAPSULE, PELLETS;ORAL | 217581-003 | Sep 7, 2023 | RX | Yes | Yes | 7,858,643 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Pf Prism Cv | XALKORI | crizotinib | CAPSULE, PELLETS;ORAL | 217581-002 | Sep 7, 2023 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pf Prism Cv | BOSULIF | bosutinib monohydrate | TABLET;ORAL | 203341-001 | Sep 4, 2012 | RX | Yes | Yes | 7,767,678*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Pf Prism Cv | XALKORI | crizotinib | CAPSULE, PELLETS;ORAL | 217581-002 | Sep 7, 2023 | RX | Yes | No | 7,230,098 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Pf Prism Cv | BOSULIF | bosutinib monohydrate | TABLET;ORAL | 203341-003 | Oct 27, 2017 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pf Prism Cv | BOSULIF | bosutinib monohydrate | TABLET;ORAL | 203341-002 | Sep 4, 2012 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Pf Prism Cv
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pf Prism Cv | RAPAMUNE | sirolimus | TABLET;ORAL | 021110-002 | Aug 22, 2002 | 5,212,155*PED | ⤷ Try a Trial |
| Pf Prism Cv | RAPAMUNE | sirolimus | SOLUTION;ORAL | 021083-001 | Sep 15, 1999 | 5,100,899*PED | ⤷ Try a Trial |
| Pf Prism Cv | TORISEL | temsirolimus | SOLUTION;INTRAVENOUS | 022088-001 | May 30, 2007 | 8,455,539*PED | ⤷ Try a Trial |
| Pf Prism Cv | BOSULIF | bosutinib monohydrate | TABLET;ORAL | 203341-003 | Oct 27, 2017 | 6,002,008 | ⤷ Try a Trial |
| Pf Prism Cv | TYGACIL | tigecycline | POWDER;INTRAVENOUS | 021821-001 | Jun 15, 2005 | RE40183 | ⤷ Try a Trial |
| Pf Prism Cv | VFEND | voriconazole | FOR SUSPENSION;ORAL | 021630-001 | Dec 19, 2003 | 5,116,844 | ⤷ Try a Trial |
| Pf Prism Cv | RAPAMUNE | sirolimus | TABLET;ORAL | 021110-002 | Aug 22, 2002 | 5,403,833*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for PF PRISM CV drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Oral Suspension | 40 mg/mL | ➤ Subscribe | 2010-10-08 |
| ➤ Subscribe | Injection | 25 mg/mL, 1.8 mL vial | ➤ Subscribe | 2011-05-25 |
| ➤ Subscribe | Tablets | 0.5 mg | ➤ Subscribe | 2010-08-25 |
| ➤ Subscribe | Extended-release Tablets | 25 mg | ➤ Subscribe | 2015-05-08 |
| ➤ Subscribe | Tablets | 1 mg and 5 mg | ➤ Subscribe | 2018-02-23 |
| ➤ Subscribe | Tablets | 400 mg | ➤ Subscribe | 2018-10-15 |
| ➤ Subscribe | Tablets | 50 mg and 200 mg | ➤ Subscribe | 2008-04-14 |
| ➤ Subscribe | For Injection | 50 mg per vial | ➤ Subscribe | 2009-06-15 |
| ➤ Subscribe | Tablets | 1 mg and 2 mg | ➤ Subscribe | 2009-12-17 |
| ➤ Subscribe | Extended-release Tablets | 50 mg and 100 mg | ➤ Subscribe | 2012-02-29 |
| ➤ Subscribe | Tablets | 0.5 mg and 1 mg | ➤ Subscribe | 2010-05-10 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Tablets | 100mg and 500mg | ➤ Subscribe | 2016-09-06 |
| ➤ Subscribe | For Injection | 200 mg/vial | ➤ Subscribe | 2008-09-12 |
International Patents for Pf Prism Cv Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 3398597 | ⤷ Try a Trial |
| South Korea | 20050026025 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 0102369 | ⤷ Try a Trial |
| Australia | 782092 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2006128150 | ⤷ Try a Trial |
| Guatemala | 200400183 | ⤷ Try a Trial |
| Taiwan | 200901989 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Pf Prism Cv Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0536515 | 2006/030 | Ireland | ⤷ Try a Trial | PRODUCT NAME: TIGECYCLINE OR A PHARMACOLOGICALLY ACCEPTABLE ORGANIC OR INORGANIC SALT OR METAL COMPLEX THEREOF; REGISTRATION NO/DATE: EU/1/06/336/001 20060424 |
| 0536515 | C300244 | Netherlands | ⤷ Try a Trial | PRODUCT: TIGECYCLINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF METAALCOMPLEX, IN HET BIJZONDER TIGECYCLINE; FIRST REGISTRATION, DATE: EU/1/06/336/001, 20060424 |
| 1218348 | CR 2013 00010 | Denmark | ⤷ Try a Trial | PRODUCT NAME: AXITINIB, EVENTUELT I FORM AF ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/12/777/001-006 20120903 |
| 1218348 | PA2013003,C1218348 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: AXITINIBUM; REGISTRATION NO/DATE: EU/1/12/777/001 - EU/1/12/777/006 20120903 |
| 1218348 | 2013C/015 | Belgium | ⤷ Try a Trial | PRODUCT NAME: AXITINIB; AUTHORISATION NUMBER AND DATE: EU/1/12/777/001 20120905 |
| 0648494 | 01C0037 | France | ⤷ Try a Trial | PRODUCT NAME: SIROLIMUS; NAT. REGISTRATION NO/DATE: EU/1/01/171/001-005 20010313; FIRST REGISTRATION: IKS55243 20000926 |
| 1666481 | 132017000095300 | Italy | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB, OPZIONALMENTE NELLA FORMA DI UN SALE FARMACEUTICAMENTE ACCETTABILE, COMPRENDENTE IL SALE CITRATO(XELJANZ); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/17/1178/001-004, 20170324 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.