Novartis Company Profile
✉ Email this page to a colleague
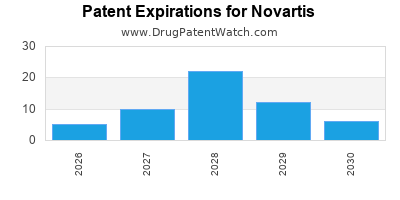
What is the competitive landscape for NOVARTIS, and when can generic versions of NOVARTIS drugs launch?
NOVARTIS has one hundred and eighty-five approved drugs.
There are one hundred and three US patents protecting NOVARTIS drugs.
There are two thousand seven hundred and forty-eight patent family members on NOVARTIS drugs in sixty-nine countries and five hundred and twenty supplementary protection certificates in nineteen countries.
Summary for Novartis
| International Patents: | 2748 |
| US Patents: | 103 |
| Tradenames: | 154 |
| Ingredients: | 139 |
| NDAs: | 185 |
| Drug Master File Entries: | 1 |
| Patent Litigation for Novartis: | See patent lawsuits for Novartis |
| PTAB Cases with Novartis as patent owner: | See PTAB cases with Novartis as patent owner |
Drugs and US Patents for Novartis
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-001 | Oct 29, 2007 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Novartis | PERCORTEN | desoxycorticosterone pivalate | INJECTABLE;INJECTION | 008822-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Novartis | EXFORGE HCT | amlodipine besylate; hydrochlorothiazide; valsartan | TABLET;ORAL | 022314-002 | Apr 30, 2009 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Novartis | TAFINLAR | dabrafenib mesylate | CAPSULE;ORAL | 202806-001 | May 29, 2013 | RX | Yes | No | 10,869,869*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Novartis | MAYZENT | siponimod | TABLET;ORAL | 209884-001 | Mar 26, 2019 | RX | Yes | No | 8,492,441 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Novartis | NEORAL | cyclosporine | CAPSULE;ORAL | 050715-003 | Jul 14, 1995 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Novartis
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novartis | EXJADE | deferasirox | TABLET, FOR SUSPENSION;ORAL | 021882-003 | Nov 2, 2005 | 6,596,750 | ⤷ Try a Trial |
| Novartis | TRILEPTAL | oxcarbazepine | SUSPENSION;ORAL | 021285-001 | May 25, 2001 | 8,119,148*PED | ⤷ Try a Trial |
| Novartis | FAMVIR | famciclovir | TABLET;ORAL | 020363-002 | Jun 29, 1994 | 5,866,581*PED | ⤷ Try a Trial |
| Novartis | SEEBRI | glycopyrrolate | POWDER;INHALATION | 207923-001 | Oct 29, 2015 | 8,580,306 | ⤷ Try a Trial |
| Novartis | LAMISIL | terbinafine hydrochloride | CREAM;TOPICAL | 020192-001 | Dec 30, 1992 | 4,755,534*PED | ⤷ Try a Trial |
| Novartis | TRASICOR | oxprenolol hydrochloride | CAPSULE;ORAL | 018166-002 | Dec 28, 1983 | 3,483,221 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for NOVARTIS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Tablets | 100 mg | ➤ Subscribe | 2005-12-30 |
| ➤ Subscribe | Tablets | 0.25 mg, 0.5 mg, and 0.75 mg | ➤ Subscribe | 2013-09-30 |
| ➤ Subscribe | Oral Solution | 2 mg/mL | ➤ Subscribe | 2004-11-05 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2014-06-18 |
| ➤ Subscribe | Tablets | 2.5 mg | ➤ Subscribe | 2006-03-02 |
| ➤ Subscribe | Capsules | 0.5 mg | ➤ Subscribe | 2014-09-22 |
| ➤ Subscribe | Tablets | 80 mg/12.5 mg, 160 mg/12.5 mg and 160 mg/25 mg | ➤ Subscribe | 2005-12-02 |
| ➤ Subscribe | Tablets | 180 mg | ➤ Subscribe | 2015-10-23 |
| ➤ Subscribe | Capsules | 400 mg | ➤ Subscribe | 2014-01-24 |
| ➤ Subscribe | Tablets | 5 mg/160 mg | ➤ Subscribe | 2007-10-22 |
| ➤ Subscribe | Capsules | 150 mg and 200 mg | ➤ Subscribe | 2013-01-29 |
| ➤ Subscribe | Tablets | 10 mg/160 mg | ➤ Subscribe | 2007-10-01 |
| ➤ Subscribe | Injection | 4 mg/100 mg, 100 mL vial | ➤ Subscribe | 2012-01-31 |
| ➤ Subscribe | Tablets | 5 mg/12.5 mg/160 mg, 5 mg/25 mg/160 mg, 10 mg/25 mg/160 mg and 10 mg/25 mg/320 m | ➤ Subscribe | 2009-09-14 |
| ➤ Subscribe | Nasal Spray | 0.665 mg/ Spray | ➤ Subscribe | 2009-06-29 |
| ➤ Subscribe | Tablets | 125 mg, 250 mg and 500 mg | ➤ Subscribe | 2004-12-28 |
| ➤ Subscribe | Oral Suspension | 300 mg/5 mL | ➤ Subscribe | 2006-12-26 |
| ➤ Subscribe | Tablets | 12.5 mg and 25 mg | ➤ Subscribe | 2014-02-04 |
| ➤ Subscribe | Delayed-release Tablets | 180 mg | ➤ Subscribe | 2009-06-04 |
| ➤ Subscribe | Tablets | 250 mg | ➤ Subscribe | 2011-03-14 |
| ➤ Subscribe | Capsules | 1.5 mg, 3 mg, 4.5 mg and 6 mg | ➤ Subscribe | 2004-04-21 |
| ➤ Subscribe | Tablets | 125 mg, 250 mg, and 500 mg | ➤ Subscribe | 2011-10-28 |
| ➤ Subscribe | Tablets for Oral Suspension | 2 mg, 3 mg and 5 mg | ➤ Subscribe | 2016-12-30 |
| ➤ Subscribe | Transdermal System Extended-release | 13.3 mg/24 hr | ➤ Subscribe | 2013-01-22 |
| ➤ Subscribe | Tablets | 2.5 mg, 5 mg, and 7.5 mg | ➤ Subscribe | 2014-12-10 |
| ➤ Subscribe | Tablets | 90 mg and 360 mg | ➤ Subscribe | 2015-10-19 |
| ➤ Subscribe | Tablets | 100 mg and 400 mg | ➤ Subscribe | 2007-03-12 |
| ➤ Subscribe | Tablets | 320 mg/12.5 mg and 320 mg/25 mg | ➤ Subscribe | 2007-02-07 |
| ➤ Subscribe | Tablets | 180 mg | ➤ Subscribe | 2016-04-28 |
| ➤ Subscribe | Capsules | 20 mg and 40 mg | ➤ Subscribe | 2008-06-04 |
| ➤ Subscribe | Tablets | 5 mg/320 mg | ➤ Subscribe | 2007-11-26 |
| ➤ Subscribe | Injection | 0.8 mg (base) /mL | ➤ Subscribe | 2008-06-11 |
| ➤ Subscribe | Tablets | 10 mg/320 mg | ➤ Subscribe | 2007-11-09 |
| ➤ Subscribe | Ophthalmic Solution | 0.003% | ➤ Subscribe | 2015-12-30 |
| ➤ Subscribe | Tablets | 10 mg/12.5 mg/160 mg | ➤ Subscribe | 2009-10-22 |
| ➤ Subscribe | Tablets | 150 mg, 300 mg and 600 mg | ➤ Subscribe | 2006-05-05 |
| ➤ Subscribe | Tablets | 50 mg and 75 mg | ➤ Subscribe | 2014-01-07 |
| ➤ Subscribe | Delayed-release Tablets | 360 mg | ➤ Subscribe | 2009-02-02 |
| ➤ Subscribe | Tablets | 60 mg and 120 mg | ➤ Subscribe | 2004-12-22 |
| ➤ Subscribe | Tablets | 40 mg, 80 mg,160 mg | ➤ Subscribe | 2004-12-28 |
International Patents for Novartis Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 2009011881 | ⤷ Try a Trial |
| China | 104306350 | ⤷ Try a Trial |
| Singapore | 189229 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2005025551 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2004094595 | ⤷ Try a Trial |
| Austria | 397445 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Novartis Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0564409 | 02C0012 | France | ⤷ Try a Trial | PRODUCT NAME: IMATINIB MESILATE; NAT. REGISTRATION NO/DATE: EU/1/01/198/001 20011107; FIRST REGISTRATION: LI - IKS 55 807 20010621 |
| 0771217 | CA 2006 00038 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL (SOM BETA-CYCLODEXTRIN-CLATHRAT) OG DROSPIRENON; NAT. REG. NO/DATE: 38687 20060627; FIRST REG. NO/DATE: EU RVG 31781 20050804 |
| 1915993 | 132013902215595 | Italy | ⤷ Try a Trial | PRODUCT NAME: ALISKIREN O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE E AMLODIPINA O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE(RASILAMLO); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/11/686/001-056, 20110414 |
| 0443983 | 97C0039 | Belgium | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN; NAT. REGISTRATION NO/DATE: 206 IS 239 F 4 19970520; FIRST REGISTRATION: DE 36983.00.00 19960513 |
| 2435025 | CR 2019 00032 | Denmark | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION AF GLYCOPYRROLAT, HERUNDER ALLE FARMACEUTISK ACCEPTABLE SALTE, ESTERE, ENANTIOMERER ELLER SOLVATER DERAF, OG FORMOTEROL, HERUNDER ALLE FARMACEUTISK ACCEPTABLE SALTE, ESTERE, ENANTIOMERE ELLER SOLVATER DERAF; REG. NO/DATE: EU/1/18/1339 20181220 |
| 0275821 | SPC/GB01/039 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ZOLEDRONIC ACID OPTIONALLY IN THE FORM OF A SALT THEREOF; NATIONAL REGISTRATION NO/DATE: EU/1/01/176/001-003 20010320; FIRST REGISTRATION: CH 55463 20001128 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.