Bristol Myers Squibb Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BRISTOL MYERS SQUIBB, and what generic alternatives to BRISTOL MYERS SQUIBB drugs are available?
BRISTOL MYERS SQUIBB has seventy approved drugs.
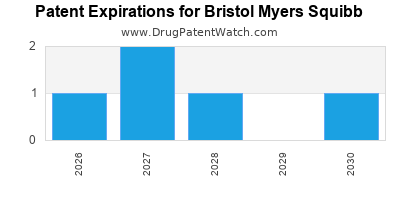
There are thirteen US patents protecting BRISTOL MYERS SQUIBB drugs.
There are five hundred and ninety-six patent family members on BRISTOL MYERS SQUIBB drugs in fifty-one countries and one hundred and forty-three supplementary protection certificates in eighteen countries.
Summary for Bristol Myers Squibb
| International Patents: | 596 |
| US Patents: | 13 |
| Tradenames: | 60 |
| Ingredients: | 55 |
| NDAs: | 70 |
Drugs and US Patents for Bristol Myers Squibb
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bristol Myers Squibb | AZACTAM | aztreonam | INJECTABLE;INJECTION | 050580-001 | Dec 31, 1986 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-001 | Jun 28, 2006 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-004 | Jun 29, 2006 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bristol Myers Squibb | IDHIFA | enasidenib mesylate | TABLET;ORAL | 209606-002 | Aug 1, 2017 | RX | Yes | Yes | 10,610,125 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bristol Myers Squibb
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bristol Myers Squibb | VIDEX | didanosine | TABLET, CHEWABLE;ORAL | 020154-006 | Oct 28, 1999 | 5,616,566*PED | ⤷ Try a Trial |
| Bristol Myers Squibb | SUSTIVA | efavirenz | CAPSULE;ORAL | 020972-002 | Sep 17, 1998 | 5,519,021*PED | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-003 | Jun 29, 2006 | 9,101,621 | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-005 | Dec 21, 2011 | 9,155,730 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for BRISTOL MYERS SQUIBB drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 20 mg, 50 mg, 70 mg and 100 mg | ➤ Subscribe | 2010-06-28 |
| ➤ Subscribe | Tablets | 80 mg and 140 mg | ➤ Subscribe | 2011-06-17 |
| ➤ Subscribe | Delayed-release Capsules | 200 mg, 250 mg and 400 mg | ➤ Subscribe | 2004-06-01 |
| ➤ Subscribe | Capsules | 5 mg, 10 mg and 15 mg | ➤ Subscribe | 2010-08-30 |
| ➤ Subscribe | Capsules | 300 mg | ➤ Subscribe | 2009-07-20 |
| ➤ Subscribe | Capsules | 200 mg | ➤ Subscribe | 2010-02-16 |
| ➤ Subscribe | Capsules | 50 mg, 100 mg and 200 mg | ➤ Subscribe | 2016-11-03 |
| ➤ Subscribe | Tablets | 0.5 mg and 1 mg | ➤ Subscribe | 2010-06-14 |
| ➤ Subscribe | Tablets | 30 mg | ➤ Subscribe | 2005-06-01 |
| ➤ Subscribe | Tablets | 80 mg and 140 mg | ➤ Subscribe | 2011-06-16 |
| ➤ Subscribe | Capsules | 25 mg | ➤ Subscribe | 2010-07-12 |
| ➤ Subscribe | Capsules | 2.5 mg and 20 mg | ➤ Subscribe | 2016-07-12 |
| ➤ Subscribe | Capsules | 100 mg and 150 mg | ➤ Subscribe | 2010-03-19 |
| ➤ Subscribe | Tablets | 600 mg | ➤ Subscribe | 2009-04-09 |
| ➤ Subscribe | Nasal Spray | 4 mg/spray | ➤ Subscribe | 2016-07-15 |
| ➤ Subscribe | Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2016-12-28 |
International Patents for Bristol Myers Squibb Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Portugal | 3257500 | ⤷ Try a Trial |
| Canada | 2727824 | ⤷ Try a Trial |
| Ukraine | 78232 | ⤷ Try a Trial |
| Canada | 2741575 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bristol Myers Squibb Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0770388 | PA2009004 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOLI VALERAS + DIENOGESTUM; NAT. REGISTRATION NO/DATE: LT/1/09/1512/001, 2009 04 06 LT/1/09/1512/002, 2009 04 06 LT/1/09/1512/003 20090406; FIRST REGISTRATION: BE 327792 20081103 |
| 1304992 | 92401 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: CLINDAMYCINE(EN TANT QUE PHOPSHATE DE CLINDAMYCINE)ET TRETINOINE |
| 0273277 | SPC/GB96/049 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: STAVUDINE; REGISTERED: UK EU/1/96/009/001 19960508; UK EU/1/96/009/002 19960508; UK EU/1/96/009/003 19960508; UK EU/1/96/009/004 19960508; UK EU/1/96/009/005 19960508; UK EU/1/96/009/006 19960508; UK EU/1/96/009/007 19960508; UK EU/1/96/009/008 19960508; UK EU/1/96/009/009 19960508 |
| 0582455 | 2008/016 | Ireland | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF EFAVIRENZ AND EMTRICTABINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTRATION NO/DATE: EU/1/07/430/001 20071213 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.