AMPHETAMINE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Amphetamine, and what generic alternatives are available?
Amphetamine is a drug marketed by Actavis Labs Fl Inc, Alkem Labs Ltd, Amneal Pharms, Aurolife Pharma Llc, Bionpharma, Cerovene Inc, Dr Reddys Labs Sa, Epic Pharma Llc, Glenmark Pharms Ltd, Granules, Lannett, Novast Labs, Prinston Inc, Rhodes Pharms, Senores Pharms, Specgx Llc, and Sun Pharm Inds Inc. and is included in seventeen NDAs.
The generic ingredient in AMPHETAMINE is amphetamine sulfate. There are fifty-five drug master file entries for this compound. Fourteen suppliers are listed for this compound. Additional details are available on the amphetamine sulfate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Amphetamine
A generic version of AMPHETAMINE was approved as amphetamine sulfate by AMNEAL PHARMS on September 26th, 2018.
Summary for AMPHETAMINE
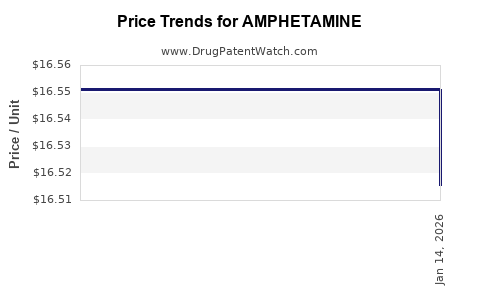
See drug prices for AMPHETAMINE
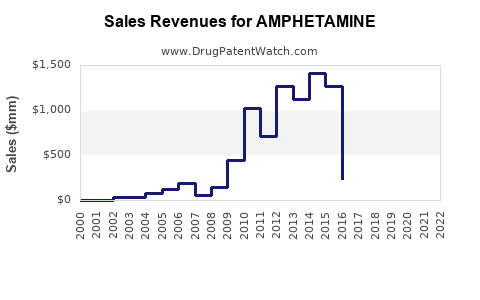
Recent Clinical Trials for AMPHETAMINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Stephen Robert Marder | Phase 2 |
| Virginia Commonwealth University | Phase 2 |
| Boston Children's Hospital | Phase 1 |
Medical Subject Heading (MeSH) Categories for AMPHETAMINE
Paragraph IV (Patent) Challenges for AMPHETAMINE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ADZENYS XR-ODT | Extended-release Orally Disintegrating Tablets | amphetamine | 3.1 mg, 6.3 mg, 9.4 mg, 12.5 mg, 15.7 mg, 18.8 mg | 204326 | 1 | 2016-05-10 |
US Patents and Regulatory Information for AMPHETAMINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rhodes Pharms | AMPHETAMINE SULFATE | amphetamine sulfate | TABLET;ORAL | 213852-002 | Sep 7, 2021 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Novast Labs | AMPHETAMINE SULFATE | amphetamine sulfate | TABLET;ORAL | 213763-002 | Aug 24, 2020 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Senores Pharms | AMPHETAMINE SULFATE | amphetamine sulfate | TABLET;ORAL | 212901-001 | May 22, 2020 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Prinston Inc | AMPHETAMINE SULFATE | amphetamine sulfate | TABLET;ORAL | 211861-001 | Mar 11, 2020 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


