OTSUKA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for OTSUKA, and what generic alternatives to OTSUKA drugs are available?
OTSUKA has seventeen approved drugs.
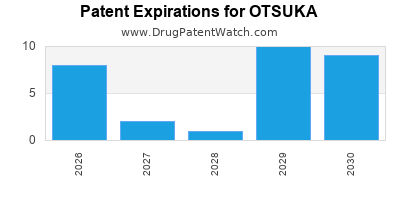
There are fifty-six US patents protecting OTSUKA drugs.
There are one thousand and seventeen patent family members on OTSUKA drugs in fifty-four countries and forty-nine supplementary protection certificates in sixteen countries.
Summary for OTSUKA
| International Patents: | 1017 |
| US Patents: | 56 |
| Tradenames: | 15 |
| Ingredients: | 10 |
| NDAs: | 17 |
| Patent Litigation for OTSUKA: | See patent lawsuits for OTSUKA |
Drugs and US Patents for OTSUKA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-005 | Nov 13, 2017 | RX | Yes | No | 8,017,615 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-001 | Nov 13, 2017 | RX | Yes | No | 11,476,952 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | REXULTI | brexpiprazole | TABLET;ORAL | 205422-005 | Jul 10, 2015 | AB | RX | Yes | No | 7,888,362 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | |
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-001 | Nov 13, 2017 | RX | Yes | No | 11,229,378 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-001 | Nov 13, 2017 | RX | Yes | No | 8,956,288 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | ABILIFY ASIMTUFII | aripiprazole | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 217006-001 | Apr 27, 2023 | RX | Yes | No | 8,338,427 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-005 | Nov 13, 2017 | RX | Yes | No | 9,149,577 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OTSUKA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-002 | Nov 13, 2017 | 8,642,760 | ⤷ Try a Trial |
| Otsuka America | MERETEK UBT KIT (W/ PRANACTIN) | urea c-13 | FOR SOLUTION;ORAL | 020586-001 | Sep 17, 1996 | 4,830,010 | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | SOLUTION;ORAL | 021713-001 | Dec 10, 2004 | 7,053,092 | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET;ORAL | 021436-003 | Nov 15, 2002 | 7,053,092 | ⤷ Try a Trial |
| Otsuka Pharm | BUSULFEX | busulfan | INJECTABLE;INJECTION | 020954-001 | Feb 4, 1999 | 5,430,057*PED | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET;ORAL | 021436-003 | Nov 15, 2002 | 9,387,182 | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET;ORAL | 021436-001 | Nov 15, 2002 | 5,006,528*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for OTSUKA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 2 mg, 5 mg, 10 mg, 15 mg, 20 mg and 30 mg | ➤ Subscribe | 2006-11-15 |
| ➤ Subscribe | Tablets | 15 mg and 30 mg | ➤ Subscribe | 2013-09-23 |
| ➤ Subscribe | Injection | 6 mg/mL | ➤ Subscribe | 2012-12-26 |
| ➤ Subscribe | Oral Solution | 1 mg/mL | ➤ Subscribe | 2007-12-20 |
| ➤ Subscribe | Orally Disintegrating Tablets | 10 mg, 15 mg, 20 mg and 30 mg | ➤ Subscribe | 2006-11-15 |
| ➤ Subscribe | Tablets | 60 mg | ➤ Subscribe | 2018-03-26 |
International Patents for OTSUKA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Australia | 2012209853 | ⤷ Try a Trial |
| Ukraine | 99476 | ⤷ Try a Trial |
| Denmark | 2497054 | ⤷ Try a Trial |
| Japan | 5143290 | ⤷ Try a Trial |
| Japan | 5730822 | ⤷ Try a Trial |
| Israel | 222396 | ⤷ Try a Trial |
| South Korea | 101475666 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for OTSUKA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2207786 | 122023000069 | Germany | ⤷ Try a Trial | PRODUCT NAME: ZUSAMMENSETZUNG UMFASSEND: CEDAZURIDIN ODER EIN PHARMAZEUTISCH AKZEPTABLES SALZ DAVON; UND DECITABIN; REGISTRATION NO/DATE: EU/1/23/1756 20230915 |
| 1675573 | C01675573/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: ARIPIPRAZOL; REGISTRATION NO/DATE: SWISSMEDIC 63177 28.04.2014 |
| 1675573 | 239 5013-2014 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: ARIPIPRAZOL; REGISTRATION NO/DATE: EU/1/13/882/001 - EU/1/13/882/004 20131119 |
| 1869025 | SPC/GB18/038 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: BREXPIPRAZOLE OR A SALT THEREOF; REGISTERED: UK EU/1/18/1294/001(NI) 20180730; UK PLGB 506970019 20180730; UK PLGB 506970020 20180730; UK PLGB 506970021 20180730; UK PLGB 506970022 20180730; UK PLGB 506970023 20180730; UK EU/1/18/1294/007(NI) 20180730; UK EU/1/18/1294/008(NI) 20180730; UK EU/1/18/1294/009(NI) 20180730; UK EU/1/18/1294/010(NI) 20180730; UK EU/1/18/1294/011(NI) 20180730; UK PLGB 506970024 20180730; UK EU/1/18/1294/002(NI) 20180730; UK EU/1/18/1294/003(NI) 20180730; UK EU/1/18/1294/004(NI) 20180730; UK EU/1/18/1294/005(NI) 20180730; UK EU/1/18/1294/006(NI) 20180730 |
| 0367141 | SPC/GB04/039 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ARIPIPRAZOLE OR A SALT THEREOF; REGISTERED: UK EU/1/04/276/001 20040604; UK EU/1/04/276/002 20040604; UK EU/1/04/276/003 20040604; UK EU/1/04/276/004 20040604; UK EU/1/04/276/005 20040604; UK EU/1/04/276/006 20040604; UK EU/1/04/276/007 20040604; UK EU/1/04/276/008 20040604; UK EU/1/04/276/009 20040604; UK EU/1/04/276/010 20040604; UK EU/1/04/276/011 20040604; UK EU/1/04/276/012 20040604; UK EU/1/04/276/013 20040604; UK EU/1/04/276/014 20040604; UK EU/1/04/276/015 20040604; UK EU/1/04/276/016 20040604; UK EU/1/04/276/017 20040604; UK EU/1/04/276/018 20040604; UK EU/1/04/276/019 20040604; UK EU/1/04/276/020 20040604 |
| 2207786 | PA2023538 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: CEDAZURIDINAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; REGISTRATION NO/DATE: EU/1/23/1756 20230915 |
| 1675573 | 92427 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: ARIPIPRAZOLE |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.