VITEKTA Drug Patent Profile
✉ Email this page to a colleague
When do Vitekta patents expire, and when can generic versions of Vitekta launch?
Vitekta is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries.
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this compound. Additional details are available on the elvitegravir profile page.
DrugPatentWatch® Generic Entry Outlook for Vitekta
Vitekta was eligible for patent challenges on August 27, 2016.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 26, 2027. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for VITEKTA
| International Patents: | 92 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 82 |
| Patent Applications: | 1,458 |
| Formulation / Manufacturing: | see details |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VITEKTA |
| DailyMed Link: | VITEKTA at DailyMed |
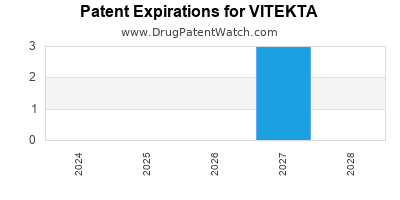
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VITEKTA
Generic Entry Date for VITEKTA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Anatomical Therapeutic Chemical (ATC) Classes for VITEKTA
US Patents and Regulatory Information for VITEKTA
VITEKTA is protected by three US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VITEKTA is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting VITEKTA
4-oxoquinoline compound and use thereof as pharmaceutical agent
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Stable crystal of 4-oxoquinoline compound
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Stable crystal of 4-oxoquinoline compound
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-001 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-002 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-001 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-001 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-002 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-002 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VITEKTA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences International Ltd | Vitekta | elvitegravir | EMEA/H/C/002577 Vitekta co-administered with a ritonavir-boosted protease inhibitor and with other antiretroviral agents, is indicated for the treatment of human-immunodeficiency-virus-1 (HIV-1) infection in adults who are infected with HIV-1 without known mutations associated with resistance to elvitegravir. |
Withdrawn | no | no | no | 2013-11-13 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VITEKTA
See the table below for patents covering VITEKTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 4299563 | COMBINAISON COMPRENANT UN CRISTAL STABLE D'UN COMPOSÉ 4-OXOQUINOLINE (COMBINATION COMPRISING A STABLE CRYSTAL OF A 4-OXOQUINOLINE COMPOUND) | ⤷ Try a Trial |
| Hong Kong | 1080852 | 4-OXOQUINOLINE COMPOUNDS AND UTILIZATION THEREOF AS HIV INTEGRASE INHIBITORS | ⤷ Try a Trial |
| European Patent Office | 3406596 | COMPOSÉ D'OXOQUINOLINE 4 ET UTILISATION ASSOCIÉE EN TANT QU'INHIBITEUR CONTRE L'INTÉGRASE DU HIV (4-OXOQUINOLINE COMPOUND AND USE THEREOF AS HIV INTEGRASE INHIBITOR) | ⤷ Try a Trial |
| China | 1956961 | Stable crystal form of 4-oxoquinoline compound | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2005113508 | ⤷ Try a Trial | |
| Austria | 443048 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VITEKTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1564210 | CA 2013 00058 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR I ENHVER FORM SOM ER OMFATTET AF GRUNDPATENTET; REG. NO/DATE: EU/1/13/830/001-002 20130524 |
| 1564210 | 489 | Finland | ⤷ Try a Trial | |
| 1564210 | 132013902209844 | Italy | ⤷ Try a Trial | PRODUCT NAME: COMPRENDENTE IL PRODOTTO ELVITEGRAVIR COME UNO DEI PRINCIPI ATTIV(STRIBILD); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/13/830/001-002, 20130524 |
| 1564210 | PA2013018 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIRUM; REGISTRATION NO/DATE: EU/1/13/830/001, 2013 05 24 EU/1/13/830/002 20130524 |
| 1564210 | 92307 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR SOUS TOUTES SES FORMES COMME PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/13/830/001-002 - STRIBILD-ELVITEGRAVIR/... 20130527 |
| 1564210 | CR 2013 00058 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR ELLER ET HYDRAT, ET SOLVAT, EN TAUTOMER ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/13/830/001-002 20130527 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


