TRULANCE Drug Patent Profile
✉ Email this page to a colleague
When do Trulance patents expire, and what generic alternatives are available?
Trulance is a drug marketed by Salix and is included in one NDA. There are nine patents protecting this drug and one Paragraph IV challenge.
This drug has sixty-eight patent family members in sixteen countries.
The generic ingredient in TRULANCE is plecanatide. One supplier is listed for this compound. Additional details are available on the plecanatide profile page.
DrugPatentWatch® Generic Entry Outlook for Trulance
Trulance was eligible for patent challenges on January 19, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 5, 2034. This may change due to patent challenges or generic licensing.
There have been eight patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for TRULANCE
| International Patents: | 68 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 15 |
| Clinical Trials: | 5 |
| Patent Applications: | 93 |
| Formulation / Manufacturing: | see details |
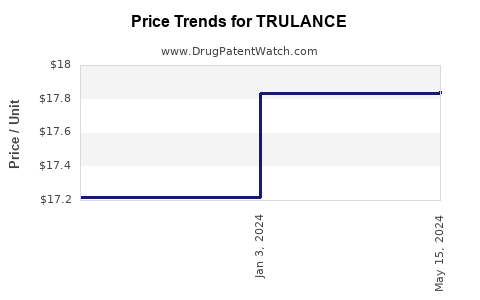
| Drug Prices: | Drug price information for TRULANCE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRULANCE |
| What excipients (inactive ingredients) are in TRULANCE? | TRULANCE excipients list |
| DailyMed Link: | TRULANCE at DailyMed |
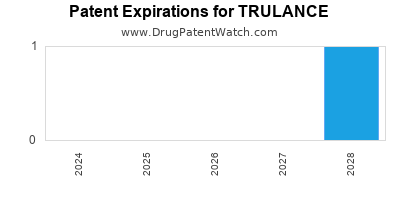
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TRULANCE
Generic Entry Date for TRULANCE*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TRULANCE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Synergy Pharmaceuticals Inc. | Phase 3 |
| Bausch Health Americas, Inc. | Phase 3 |
| Synergy Pharmaceuticals Inc. | Phase 2 |
Pharmacology for TRULANCE
| Drug Class | Guanylate Cyclase-C Agonist |
| Mechanism of Action | Guanylate Cyclase Activators |
Anatomical Therapeutic Chemical (ATC) Classes for TRULANCE
Paragraph IV (Patent) Challenges for TRULANCE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TRULANCE | Tablets | plecanatide | 3 mg | 208745 | 2 | 2021-01-19 |
US Patents and Regulatory Information for TRULANCE
TRULANCE is protected by eleven US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TRULANCE is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting TRULANCE
Ultra-pure agonists of guanylate cyclase C, method of making and using same
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Ultra-pure agonists of guanylate cyclase C, method of making and using same
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Ultra-pure agonists of guanylate cyclase C, method of making and using same
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Guanylate cyclase receptor agonists for the treatment of tissue inflammation and carcinogenesis
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Formulations of guanylate cyclase C agonists and methods of use
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: CHRONIC IDIOPATHIC CONSTIPATION
Formulations of guanylate cyclase C agonists and methods of use
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: IRRITABLE BOWEL SYNDROME WITH CONSTIPATION
Formulations of guanylate cyclase C agonists and methods of use
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Formulations of guanylate cyclase C agonists and methods of use
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: IRRITABLE BOWEL SYNDROME WITH CONSTIPATION
Formulations of guanylate cyclase C agonists and methods of use
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: CHRONIC IDIOPATHIC CONSTIPATION
Formulations of guanylate cyclase C agonists and methods of use
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TRULANCE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TRULANCE
When does loss-of-exclusivity occur for TRULANCE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 14274812
Estimated Expiration: ⤷ Try a Trial
Patent: 18226473
Estimated Expiration: ⤷ Try a Trial
Patent: 20205349
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 2015030326
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 13737
Estimated Expiration: ⤷ Try a Trial
China
Patent: 5764916
Estimated Expiration: ⤷ Try a Trial
Patent: 3388007
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 1592263
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 04138
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 21959
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 2939
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 06491
Estimated Expiration: ⤷ Try a Trial
Patent: 16522216
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 2272746
Estimated Expiration: ⤷ Try a Trial
Patent: 160039577
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TRULANCE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hong Kong | 1204454 | 鳥苷酸環化酶 激動劑的製劑及它們的使用方法 (FORMULATIONS OF GUANYLATE YCLASE AGONISTS AND METHODS OF USE) | ⤷ Try a Trial |
| Eurasian Patent Organization | 201592263 | УЛЬТРАЧИСТЫЕ АГОНИСТЫ ГУАНИЛАТЦИКЛАЗЫ C, СПОСОБ ИХ ПОЛУЧЕНИЯ И ИСПОЛЬЗОВАНИЯ | ⤷ Try a Trial |
| Germany | 60233040 | ⤷ Try a Trial | |
| Portugal | 1379224 | ⤷ Try a Trial | |
| European Patent Office | 1379224 | AGONISTES DU RECEPTEUR DU TYPE GUANYLATE CYCLASE SERVANT AU TRAITEMENT DE L'INFLAMMATION TISSULAIRE ET DE LA CARCINOGENESE (GUANYLATE CYCLASE RECEPTOR AGONISTS FOR THE TREATMENT OF TISSUE INFLAMMATION AND CARCINOGENESIS) | ⤷ Try a Trial |
| Canada | 2795396 | AGONISTES DU RECEPTEUR DU TYPE GUANYLATE CYCLASE POUR STIMULER LE TRANSPORT D'EAU DANS LE TRACTUS GASTRO-INTESTINAL POUR TRAITER L'INFLAMMATION DES TISSUS (GUANYLATE CYCLASE RECEPTOR AGONISTS FOR STIMULATING WATER TRANSPORT IN THE GASTROINTESTINAL TRACT TO TREAT TISSUE INFLAMMATION) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


