MAXZIDE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Maxzide, and what generic alternatives are available?
Maxzide is a drug marketed by Aurobindo Pharma Usa and is included in one NDA.
The generic ingredient in MAXZIDE is hydrochlorothiazide; triamterene. There are thirty-two drug master file entries for this compound. Twenty-nine suppliers are listed for this compound. Additional details are available on the hydrochlorothiazide; triamterene profile page.
Summary for MAXZIDE
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 134 |
| Clinical Trials: | 1 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for MAXZIDE |
| DailyMed Link: | MAXZIDE at DailyMed |

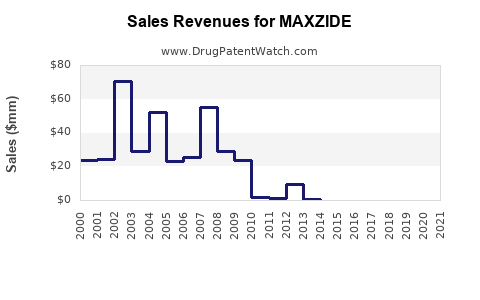
Recent Clinical Trials for MAXZIDE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| VA Office of Research and Development | |
| US Department of Veterans Affairs |
Anatomical Therapeutic Chemical (ATC) Classes for MAXZIDE
US Patents and Regulatory Information for MAXZIDE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aurobindo Pharma Usa | MAXZIDE | hydrochlorothiazide; triamterene | TABLET;ORAL | 019129-001 | Oct 22, 1984 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Aurobindo Pharma Usa | MAXZIDE-25 | hydrochlorothiazide; triamterene | TABLET;ORAL | 019129-003 | May 13, 1988 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MAXZIDE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Aurobindo Pharma Usa | MAXZIDE | hydrochlorothiazide; triamterene | TABLET;ORAL | 019129-001 | Oct 22, 1984 | ⤷ Try a Trial | ⤷ Try a Trial |
| Aurobindo Pharma Usa | MAXZIDE | hydrochlorothiazide; triamterene | TABLET;ORAL | 019129-001 | Oct 22, 1984 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for MAXZIDE
See the table below for patents covering MAXZIDE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Germany | 3381345 | ⤷ Try a Trial | |
| Belgium | 858404 | ⤷ Try a Trial | |
| Japan | H0231053 | ⤷ Try a Trial | |
| Philippines | 20531 | PHARMACEUTICAL COMBINATION COMPOSITION AND ASSOCIATED METHOD | ⤷ Try a Trial |
| Australia | 2858377 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MAXZIDE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0502314 | C300095 | Netherlands | ⤷ Try a Trial | PRODCUT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FYSIOLOGISCH VERDRAAGBAAR ZOUT, EN HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/02/213/001-010 20020419 |
| 0443983 | C00443983/03 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN + AMLODIPINE + HYDROCHLOROTHIAZIDE; REGISTRATION NUMBER/DATE: SWISSMEDIC 59407 16.09.2009 |
| 0503785 | CA 2011 00026 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF OLMESARTAN MEDOXOMIL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND AMLODIPINE BESYLATE AND HYDROCHLOROTHIAZIDE; NAT. REG. NO/DATE: 46260-46269 (DK) 20110323; FIRST REG. NO/DATE: DE 79810.00.00 20101216 |
| 0480717 | 98C0025 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LOSARTAN POTASSIUM; HYDROCHLOROTHIAZIDE; NAT. REGISTRATION NO/DATE: NL 20 037 19950215; FIRST REGISTRATION: FR - NL 20 037 19950215 |
| 0454511 | 99C0009 | Belgium | ⤷ Try a Trial | PRODUCT NAME: IRBESARTAN / HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/98/086/001 19981015 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


