Noden Pharma Company Profile
✉ Email this page to a colleague
What is the competitive landscape for NODEN PHARMA, and when can generic versions of NODEN PHARMA drugs launch?
NODEN PHARMA has four approved drugs.
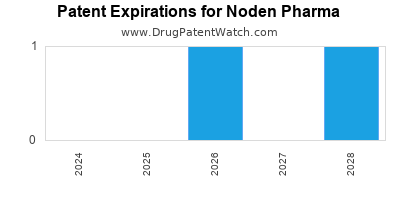
There are fourteen US patents protecting NODEN PHARMA drugs.
There are seventy-two patent family members on NODEN PHARMA drugs in twenty-six countries and thirty-nine supplementary protection certificates in fourteen countries.
Drugs and US Patents for Noden Pharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Noden Pharma | GOPRELTO | cocaine hydrochloride | SOLUTION;NASAL | 209963-001 | Dec 14, 2017 | RX | Yes | Yes | 10,987,347 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-004 | Jan 18, 2008 | DISCN | Yes | No | 8,618,172 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-002 | Jan 18, 2008 | DISCN | Yes | No | 8,618,172 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Noden Pharma | GOPRELTO | cocaine hydrochloride | SOLUTION;NASAL | 209963-001 | Dec 14, 2017 | RX | Yes | Yes | 10,231,961 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Noden Pharma | TEKTURNA | aliskiren hemifumarate | TABLET;ORAL | 021985-002 | Mar 5, 2007 | AB | RX | Yes | Yes | 8,617,595*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Noden Pharma | TEKTURNA | aliskiren hemifumarate | TABLET;ORAL | 021985-001 | Mar 5, 2007 | AB | RX | Yes | No | 8,617,595*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Noden Pharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-003 | Jan 18, 2008 | 5,559,111*PED | ⤷ Try a Trial |
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-001 | Jan 18, 2008 | 5,559,111*PED | ⤷ Try a Trial |
| Noden Pharma | TEKTURNA | aliskiren hemifumarate | TABLET;ORAL | 021985-001 | Mar 5, 2007 | 5,559,111*PED | ⤷ Try a Trial |
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-004 | Jan 18, 2008 | 9,023,893 | ⤷ Try a Trial |
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-002 | Jan 18, 2008 | 9,023,893 | ⤷ Try a Trial |
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-004 | Jan 18, 2008 | 5,559,111*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for NODEN PHARMA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 150 mg/12.5 mg, 150 mg/25 mg, 300 mg/12.5 mg, 300 mg/25 mg | ➤ Subscribe | 2014-03-07 |
| ➤ Subscribe | Tablets | 150 mg and 300 mg | ➤ Subscribe | 2013-12-13 |
International Patents for Noden Pharma Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Taiwan | I406656 | ⤷ Try a Trial |
| Japan | 5362556 | ⤷ Try a Trial |
| Ecuador | SP066807 | ⤷ Try a Trial |
| South Korea | 101353736 | ⤷ Try a Trial |
| South Korea | 20070006776 | ⤷ Try a Trial |
| South Korea | 101442272 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Noden Pharma Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0678503 | C00678503/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: ALISKIREN; REGISTRATION NUMBER/DATE: SWISSMEDIC 58050 29.06.2007 |
| 1507558 | 12C0033 | France | ⤷ Try a Trial | PRODUCT NAME: ALISKIRENE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE E CELUI-CI, AMLODIPINE OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI, ET HYDROCHLOROTHIAZIDE OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; NAT. REGISTRATION NO/DATE: EU/1/11/730/001 20111122; FIRST REGISTRATION: CH - 6167801 20110705 |
| 1507558 | 1290018-9 | Sweden | ⤷ Try a Trial | PRODUCT NAME: ALISKIREN ELLER ETT FARMACEUTISKT ACCEPTABELT SALT DAERAV, AMLODIPIN ELLER ETT FARMACEUTISKT ACCEPTABELT SALT DAERAV, OCH HYDROKLORTIAZID ELLER ETT FARMACEUTISKT ACCEPTABELT SALT DAERAV; NAT. REGISTRATIPON NO/DATE: EU/1/11/730 20111122; FIRST REGISTRATION: CH 61678 01-05 20110705 |
| 1507558 | CR 2012 00018 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ALISKIREN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, AMLODIPIN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF OG HYDROCHLORTHIAZID ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; NAT. REG. NO/DATE: EU/1/11/730/001-060 20111122; FIRST REG. NO/DATE: CH 61678 01-05 20110705 |
| 1507558 | 113 5008-2012 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: ALISKIREN / AMLODIPIN / HYDROCHLORTIAZID; NAT. REGISTRATION NO/DATE: EU/1/11/730/001 - EU/1/11/730/060 20111122; FIRST REGISTRATION: CH 61678 01 - 61678 05 20110705 |
| 1507558 | C300528 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN ALISKIREN OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, AMLODIPINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN EN HYDROCHLOORTHIAZIDE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; NATL. REGISTRATION NO/DATE: EU/1/11/730/001-060 20111122; FIRST REGISTRATION: CH 61678 01-05 20110705 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.