Mallinckrodt Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for MALLINCKRODT INC, and when can generic versions of MALLINCKRODT INC drugs launch?
MALLINCKRODT INC has four approved drugs.
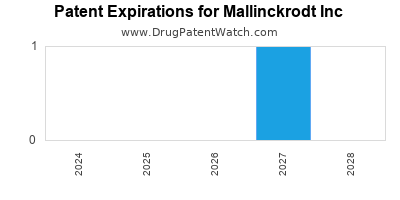
There are twelve US patents protecting MALLINCKRODT INC drugs.
There are thirty-one patent family members on MALLINCKRODT INC drugs in thirteen countries and sixty-seven supplementary protection certificates in eight countries.
Summary for Mallinckrodt Inc
| International Patents: | 31 |
| US Patents: | 12 |
| Tradenames: | 4 |
| Ingredients: | 4 |
| NDAs: | 4 |
| Drug Master File Entries: | 67 |
Drugs and US Patents for Mallinckrodt Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mallinckrodt Inc | METHADONE HYDROCHLORIDE | methadone hydrochloride | POWDER;FOR RX COMPOUNDING | 006383-003 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Mallinckrodt Inc | XARTEMIS XR | acetaminophen; oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 204031-001 | Mar 11, 2014 | DISCN | Yes | No | 8,980,319 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Mallinckrodt Inc | BAROS | sodium bicarbonate; tartaric acid | GRANULE, EFFERVESCENT;ORAL | 018509-001 | Aug 7, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Mallinckrodt Inc | METHADONE HYDROCHLORIDE | methadone hydrochloride | POWDER;FOR RX COMPOUNDING | 006383-004 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Mallinckrodt Inc | XARTEMIS XR | acetaminophen; oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 204031-001 | Mar 11, 2014 | DISCN | Yes | No | 9,050,335 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Mallinckrodt Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Mallinckrodt Inc | XARTEMIS XR | acetaminophen; oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 204031-001 | Mar 11, 2014 | 6,488,962 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for MALLINCKRODT INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Tablets | 7.5 mg/325 mg | ➤ Subscribe | 2014-04-03 |
Premature patent expirations for MALLINCKRODT INC
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for Mallinckrodt Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Brazil | 112013015622 | ⤷ Try a Trial |
| Japan | 2005507000 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2009114649 | ⤷ Try a Trial |
| South Africa | 201303988 | ⤷ Try a Trial |
| Australia | 2011345329 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Mallinckrodt Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2203431 | 2015/009 | Ireland | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REGISTRATION NO/DATE: EU/1/14/983 20150115 |
| 1948158 | 16C0018 | France | ⤷ Try a Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN,SOUS FORME DE COMPLEXE SODIQUE SACUBITRIL VALSARTAN,C'EST-A-DIRE DE (3-((1S,3R)-1-BIPHENYL-4-YLMETHYL-3-ETHOXYCARBONYL-1-BUTYLCARBAMOYL) PROPIONATE-(S)-3'-METHYL-2'-(PENTANOY(2''-(TETRAZOL-5-YLATE)BIPHENYL-4'-YLMETHYL)AMINO)BUTYRATE)DE TRISODIUM HEMIPENTAHYDRATE; REGISTRATION NO/DATE: EU/1/15/1058 20151123 |
| 2666774 | CA 2020 00037 | Denmark | ⤷ Try a Trial | PRODUCT NAME: RELEBACTAM, OPTIONALLY IN THE FORM OF THE MONOHYDRATE, IMIPENEM AND CILASTATIN, OPTIONALLY IN THE FORM OF THE SODIUM SALT; REG. NO/DATE: EU/1/19/1420 20200217 |
| 1713823 | 1490064-1 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SIMEPREVIR, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING SIMEPREVIR SODIUM; REG. NO/DATE: EU/1/14/924 20140516 |
| 2822954 | 18C1035 | France | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE,EN PARTICULIER BICTEGRAVIR DE SODIUM; REGISTRATION NO/DATE: EU/1/18/1289 20180625 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.