Bristol Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BRISTOL, and what generic alternatives to BRISTOL drugs are available?
BRISTOL has one hundred and sixteen approved drugs.
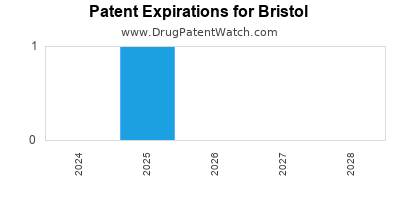
There are fifty-five US patents protecting BRISTOL drugs.
There are one thousand six hundred and ninety-one patent family members on BRISTOL drugs in fifty-eight countries and three hundred and nine supplementary protection certificates in nineteen countries.
Summary for Bristol
| International Patents: | 1691 |
| US Patents: | 55 |
| Tradenames: | 93 |
| Ingredients: | 81 |
| NDAs: | 116 |
| Drug Master File Entries: | 1 |
| Patent Litigation for Bristol: | See patent lawsuits for Bristol |
| PTAB Cases with Bristol as patent owner: | See PTAB cases with Bristol as patent owner |
Drugs and US Patents for Bristol
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bristol | VIDEX | didanosine | FOR SOLUTION;ORAL | 020156-001 | Oct 9, 1991 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers | QUESTRAN | cholestyramine | POWDER;ORAL | 016640-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | IDHIFA | enasidenib mesylate | TABLET;ORAL | 209606-002 | Aug 1, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol | POMALYST | pomalidomide | CAPSULE;ORAL | 204026-002 | Feb 8, 2013 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bristol
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bristol-myers | ABRAXANE | paclitaxel | POWDER;INTRAVENOUS | 021660-001 | Jan 7, 2005 | 6,537,579 | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-006 | Jun 5, 2013 | 6,908,432 | ⤷ Try a Trial |
| Bristol | POMALYST | pomalidomide | CAPSULE;ORAL | 204026-003 | Feb 8, 2013 | 6,315,720 | ⤷ Try a Trial |
| Bristol-myers | THALOMID | thalidomide | CAPSULE;ORAL | 020785-002 | Jan 17, 2003 | 6,235,756 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for BRISTOL drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Delayed-release Capsules | 200 mg, 250 mg and 400 mg | ➤ Subscribe | 2004-06-01 |
| ➤ Subscribe | Capsules | 200 mg | ➤ Subscribe | 2006-09-25 |
| ➤ Subscribe | Capsules | 5 mg, 10 mg and 15 mg | ➤ Subscribe | 2010-08-30 |
| ➤ Subscribe | For Injection Suspension | 100 mg/vial | ➤ Subscribe | 2015-12-11 |
| ➤ Subscribe | Capsules | 300 mg | ➤ Subscribe | 2009-07-20 |
| ➤ Subscribe | Capsules | 200 mg | ➤ Subscribe | 2010-02-16 |
| ➤ Subscribe | Capsules | 50 mg, 100 mg and 200 mg | ➤ Subscribe | 2016-11-03 |
| ➤ Subscribe | Tablets | 0.5 mg and 1 mg | ➤ Subscribe | 2010-06-14 |
| ➤ Subscribe | Tablets | 30 mg | ➤ Subscribe | 2005-06-01 |
| ➤ Subscribe | Tablets | 80 mg and 140 mg | ➤ Subscribe | 2011-06-16 |
| ➤ Subscribe | Capsules | 1 mg, 2 mg, 3 mg and 4 mg | ➤ Subscribe | 2017-02-08 |
| ➤ Subscribe | Capsules | 50 mg and 100 mg | ➤ Subscribe | 2006-12-18 |
| ➤ Subscribe | Capsules | 25 mg | ➤ Subscribe | 2010-07-12 |
| ➤ Subscribe | Capsules | 150 mg | ➤ Subscribe | 2014-02-03 |
| ➤ Subscribe | Capsules | 2.5 mg and 20 mg | ➤ Subscribe | 2016-07-12 |
| ➤ Subscribe | Injection | 10 mg/vial | ➤ Subscribe | 2013-11-05 |
| ➤ Subscribe | Capsules | 100 mg and 150 mg | ➤ Subscribe | 2010-03-19 |
| ➤ Subscribe | Tablets | 600 mg | ➤ Subscribe | 2009-04-09 |
| ➤ Subscribe | Nasal Spray | 4 mg/spray | ➤ Subscribe | 2016-07-15 |
| ➤ Subscribe | Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2016-12-28 |
| ➤ Subscribe | Tablets | 20 mg, 50 mg, 70 mg and 100 mg | ➤ Subscribe | 2010-06-28 |
| ➤ Subscribe | Tablets | 80 mg and 140 mg | ➤ Subscribe | 2011-06-17 |
International Patents for Bristol Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Eurasian Patent Organization | 201071173 | ⤷ Try a Trial |
| European Patent Office | 2097078 | ⤷ Try a Trial |
| Taiwan | 201040142 | ⤷ Try a Trial |
| Serbia | 57253 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bristol Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2049522 | 300713 | Netherlands | ⤷ Try a Trial | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), CHANGE OF OWNER(S) NAME |
| 0961612 | 2009C/046 | Belgium | ⤷ Try a Trial | PRODUCT NAME: PACLITAXEL ALBUMIN; AUTHORISATION NUMBER AND DATE: EU/1/07/428/001 20080111 |
| 0900210 | C00900210/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: ATAZANAVIR; REGISTRATION NUMBER/DATE: SWISSMEDIC 56288 06.05.2004 |
| 0582455 | SPC/GB00/035 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: EFAVIRENZ, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT; REGISTERED: CH IKS-54 908 01 19981120; CH IKS-54 908 02 19981120; CH IKS-54 908 03 19981120; UK EU/1/99/110/001 19990528; UK EU/1/99/110/002 19990528; UK EU/1/99/110/003 19990528; UK EU/1/99/110/004 19990528; UK EU/1/99/111/001 19990528; UK EU/1/99/111/002 19990528; UK EU/1/99/111/003 19990528; UK EU/1/99/111/004 19990528 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.