Boehringer Ingelheim Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BOEHRINGER INGELHEIM, and what generic alternatives to BOEHRINGER INGELHEIM drugs are available?
BOEHRINGER INGELHEIM has forty-eight approved drugs.
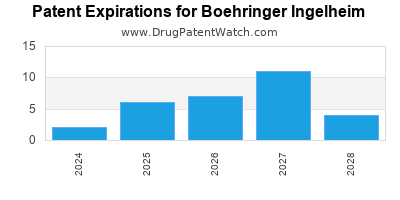
There are seventy US patents protecting BOEHRINGER INGELHEIM drugs.
There are one thousand eight hundred and twenty-six patent family members on BOEHRINGER INGELHEIM drugs in sixty-three countries and two hundred and seventeen supplementary protection certificates in nineteen countries.
Summary for Boehringer Ingelheim
| International Patents: | 1826 |
| US Patents: | 70 |
| Tradenames: | 37 |
| Ingredients: | 29 |
| NDAs: | 48 |
| Patent Litigation for Boehringer Ingelheim: | See patent lawsuits for Boehringer Ingelheim |
| PTAB Cases with Boehringer Ingelheim as petitioner: | See PTAB cases with Boehringer Ingelheim as petitioner |
Drugs and US Patents for Boehringer Ingelheim
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | JENTADUETO | linagliptin; metformin hydrochloride | TABLET;ORAL | 201281-001 | Jan 30, 2012 | AB | RX | Yes | No | 10,022,379*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | AB | RX | Yes | Yes | 11,033,552*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Boehringer Ingelheim | PRADAXA | dabigatran etexilate mesylate | PELLETS;ORAL | 214358-003 | Jun 21, 2021 | RX | Yes | No | 7,932,273*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Boehringer Ingelheim | MOBIC | meloxicam | TABLET;ORAL | 020938-002 | Aug 23, 2000 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Boehringer Ingelheim
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | SPIRIVA RESPIMAT | tiotropium bromide | SPRAY, METERED;INHALATION | 021936-001 | Sep 24, 2014 | 7,246,615*PED | ⤷ Try a Trial |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-001 | Nov 10, 1998 | 5,591,762 | ⤷ Try a Trial |
| Boehringer Ingelheim | JENTADUETO | linagliptin; metformin hydrochloride | TABLET;ORAL | 201281-002 | Jan 30, 2012 | 6,890,898 | ⤷ Try a Trial |
| Boehringer Ingelheim | STRIVERDI RESPIMAT | olodaterol hydrochloride | SPRAY, METERED;INHALATION | 203108-001 | Jul 31, 2014 | 6,846,413 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for BOEHRINGER INGELHEIM drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 10 mg and 25 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 20 mg, 40 mg and 80 mg | ➤ Subscribe | 2006-12-26 |
| ➤ Subscribe | Tablets | 80 mg/25 mg | ➤ Subscribe | 2009-02-27 |
| ➤ Subscribe | Capsules | 100 mg and 150 mg | ➤ Subscribe | 2018-10-15 |
| ➤ Subscribe | Tablets | 0.25 mg | ➤ Subscribe | 2005-05-27 |
| ➤ Subscribe | Capsules | eq. to 110 mg base | ➤ Subscribe | 2015-12-15 |
| ➤ Subscribe | Oral Suspension | 7.5 mg/5 mL | ➤ Subscribe | 2009-12-17 |
| ➤ Subscribe | Extended-releaseTablets | 2.5 mg/1000 mg 5 mg/1000 mg | ➤ Subscribe | 2018-03-28 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2015-05-04 |
| ➤ Subscribe | Extended-release Tablets | 2.25 mg and 3.75 mg | ➤ Subscribe | 2011-07-26 |
| ➤ Subscribe | Extended-release Tablets | 400 mg | ➤ Subscribe | 2013-06-21 |
| ➤ Subscribe | Tablets | 10 mg/5 mg and 25 mg/5 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 2.5 mg/500 mg, 2.5 mg/850 mg, 2.5 mg/1000 mg | ➤ Subscribe | 2015-05-04 |
| ➤ Subscribe | Tablets | 80 mg/12.5 mg and 40 mg/12.5 mg | ➤ Subscribe | 2008-12-31 |
| ➤ Subscribe | Extended-release Capsules | 25 mg and 200 mg | ➤ Subscribe | 2007-02-01 |
| ➤ Subscribe | Tablets | 0.125 mg, 0.5 mg, 1 mg and 1.5 mg | ➤ Subscribe | 2005-06-24 |
| ➤ Subscribe | Capsules | eq. to 75 mg base and 150 mg base | ➤ Subscribe | 2014-10-20 |
| ➤ Subscribe | Tablets | 0.75 mg | ➤ Subscribe | 2008-07-31 |
| ➤ Subscribe | Inhalation Powder Capsules | 18 mcg | ➤ Subscribe | 2018-05-11 |
| ➤ Subscribe | Tablets | 5 mg/500 mg5 mg/1000 mg12.5 mg/500 mg12.5 mg/1000 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Extended-release Tablets | 0.375 mg, 0.75 mg, 1.5 mg, 3 mg and 4.5 mg | ➤ Subscribe | 2010-06-01 |
| ➤ Subscribe | Extended-release Tablets | 5 mg/1000 mg10 mg/1000 mg12.5 mg/1000 mg25 mg/1000 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 20 mg, 30 mg and 40 mg | ➤ Subscribe | 2017-07-12 |
International Patents for Boehringer Ingelheim Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 2012003102 | ⤷ Try a Trial |
| Hong Kong | 1145806 | ⤷ Try a Trial |
| Colombia | 6280467 | ⤷ Try a Trial |
| Japan | 6662970 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Boehringer Ingelheim Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1532149 | PA2012022 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: LINAGLIPTINUM + METFORMINI HYDROCHLORIDUM; REGISTRATION NO/DATE: EU/1/12/780/001 - EU/1/12/780/034 20120720 |
| 1224170 | C01224170/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: NINTEDANIB; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 65330 13.08.2015 |
| 2187879 | C20170016 00337 | Estonia | ⤷ Try a Trial | PRODUCT NAME: EMPAGLIFLOSIIN/LINAGLIPTIIN;REG NO/DATE: EU/1/16/1146 15.11.2016 |
| 1562603 | C 2014 025 | Romania | ⤷ Try a Trial | PRODUCT NAME: OLODATEROL, IZOMERI OPTICI INDIVIDUALI AI ACESTUIA,AMESTECURI DE ENANTIOMERI INDIVIDUALI SAU RACEMATI, SARURILE DE ADITIE ACIDA ALE ACESTORA CU ACIZI ACCEPTABILIFARMACOLOGIC, CA SI SOLVATI SI/SAU HIDRATI AI ACESTORA, IN SPECIAL OLODATEROL SI CLORHI DRAT DE OLODATEROL -6-HIDROXI-2-{[1-(4-METOXIFENIL)-1,1-DIMETILETIL]AMINO}ETIL]-2H-1,4-BENZOXAZIN-3(4H)-ONA(CACLORHIDRAT); NATIONAL AUTHORISATION NUMBER: 6414/2014/01, 6414/2014/02, 6414/2014/03, 6414/2014/04; DATE OF NATIONAL AUTHORISATION: 20140507; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): MA211/00401; DATE OF FIRST AUTHORISATION IN EEA: 20130918 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.