Astrazeneca Ab Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ASTRAZENECA AB, and when can generic versions of ASTRAZENECA AB drugs launch?
ASTRAZENECA AB has twelve approved drugs.
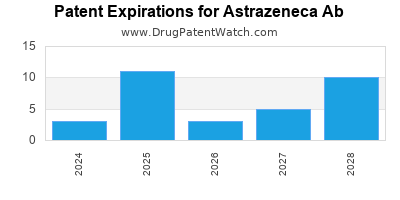
There are fifty-six US patents protecting ASTRAZENECA AB drugs. There is one tentative approval on ASTRAZENECA AB drugs.
There are eight hundred and ninety-eight patent family members on ASTRAZENECA AB drugs in fifty-four countries and one hundred and forty-two supplementary protection certificates in nineteen countries.
Summary for Astrazeneca Ab
| International Patents: | 898 |
| US Patents: | 56 |
| Tradenames: | 13 |
| Ingredients: | 10 |
| NDAs: | 12 |
| Drug Master File Entries: | 2 |
| Patent Litigation for Astrazeneca Ab: | See patent lawsuits for Astrazeneca Ab |
| PTAB Cases with Astrazeneca Ab as patent owner: | See PTAB cases with Astrazeneca Ab as patent owner |
Drugs and US Patents for Astrazeneca Ab
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | XIGDUO XR | dapagliflozin; metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 205649-002 | Oct 29, 2014 | RX | Yes | No | 9,616,028 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | RX | Yes | Yes | 8,721,615 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Astrazeneca Ab | BYETTA | exenatide synthetic | INJECTABLE;SUBCUTANEOUS | 021773-001 | Apr 28, 2005 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Astrazeneca Ab | BYDUREON PEN | exenatide synthetic | FOR SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 022200-002 | Feb 28, 2014 | DISCN | Yes | No | 8,758,292*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Astrazeneca Ab | XIGDUO XR | dapagliflozin; metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 205649-001 | Oct 29, 2014 | RX | Yes | No | 7,919,598 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | RX | Yes | No | 8,221,786 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Astrazeneca Ab
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | BYDUREON PEN | exenatide synthetic | FOR SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 022200-002 | Feb 28, 2014 | 6,479,065 | ⤷ Try a Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 9,238,076 | ⤷ Try a Trial |
| Astrazeneca Ab | XIGDUO XR | dapagliflozin; metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 205649-003 | Oct 29, 2014 | 6,936,590 | ⤷ Try a Trial |
| Astrazeneca Ab | BYETTA | exenatide synthetic | INJECTABLE;SUBCUTANEOUS | 021773-001 | Apr 28, 2005 | 7,297,761 | ⤷ Try a Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 9,198,925 | ⤷ Try a Trial |
| Astrazeneca Ab | ONGLYZA | saxagliptin hydrochloride | TABLET;ORAL | 022350-001 | Jul 31, 2009 | RE44186 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ASTRAZENECA AB drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | HydrochlorideExtended-release Tablets | 2.5 mg/1000 mg | ➤ Subscribe | 2018-10-29 |
| ➤ Subscribe | Extended-release Tablets | 5 mg/500 mg, 2.5 mg/1000 mg, and 5 mg/1000 mg | ➤ Subscribe | 2013-07-31 |
| ➤ Subscribe | Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2013-07-31 |
| ➤ Subscribe | Injection | 250 mg/mL, 1.2 mL and 2.4 mL prefilled syringe | ➤ Subscribe | 2014-06-11 |
International Patents for Astrazeneca Ab Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 4155403 | ⤷ Try a Trial |
| Japan | 2022017514 | ⤷ Try a Trial |
| European Patent Office | 3106149 | ⤷ Try a Trial |
| Brazil | PI0918904 | ⤷ Try a Trial |
| Eurasian Patent Organization | 020299 | ⤷ Try a Trial |
| Serbia | 53814 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Astrazeneca Ab Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2498758 | 132020000000034 | Italy | ⤷ Try a Trial | PRODUCT NAME: METFORMINA O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE; SAXAGLIPTIN O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE; DAPAGLIFLOZIN O UN SUO SOLVATO FARMACEUTICAMENTE ACCETTABILE.(QTRILMET); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/19/1401, 20191113 |
| 1261586 | 1290013-0 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SAXAGLIPTIN/METFORMIN; REG. NO/DATE: EU/1/11/731/001 20111124 |
| 1506211 | 92496 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE DAPAGLIFLOZINE OU D UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI ET DE METFORMINE OU D UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI,TELLE QUE PROTEGEE PAR LE BREVET DE BASE EP1506211 B1 |
| 2435025 | PA2019014,C2435025 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: GLIKOPIRONIO BROMIDAS/FORMOTEROLIS; REGISTRATION NO/DATE: EU/1/18/1339 20181218 |
| 2139494 | PA2020522 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: SASAGLIPTINAS IR DAPAGLIFLOZINAS; REGISTRATION NO/DATE: EU/1/16/1108 20160715 |
| 1261586 | SPC/GB10/001 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: SAXAGLIPTIN AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF INCLUDING SAXAGLIPTIN HYDROCHLORIDE; REGISTERED: UK EU/1/09/545/001 20091001; UK EU/1/09/545/002 20091001; UK EU/1/09/545/003 20091001; UK EU/1/09/545/004 20091001; UK EU/1/09/545/005 20091001; UK EU/1/09/545/006 20091001; UK EU/1/09/545/007 20091001; UK EU/1/09/545/008 20091001; UK EU/1/09/545/009 20091001; UK EU/1/09/545/010 20091001 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.