KLISYRI Drug Patent Profile
✉ Email this page to a colleague
When do Klisyri patents expire, and when can generic versions of Klisyri launch?
Klisyri is a drug marketed by Almirall and is included in one NDA. There are eight patents protecting this drug.
This drug has one hundred and six patent family members in twenty-six countries.
The generic ingredient in KLISYRI is tirbanibulin. One supplier is listed for this compound. Additional details are available on the tirbanibulin profile page.
DrugPatentWatch® Generic Entry Outlook for Klisyri
Klisyri will be eligible for patent challenges on December 14, 2024. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be September 7, 2038. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for KLISYRI
| International Patents: | 106 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 65 |
| Patent Applications: | 133 |
| Drug Prices: | Drug price information for KLISYRI |
| What excipients (inactive ingredients) are in KLISYRI? | KLISYRI excipients list |
| DailyMed Link: | KLISYRI at DailyMed |
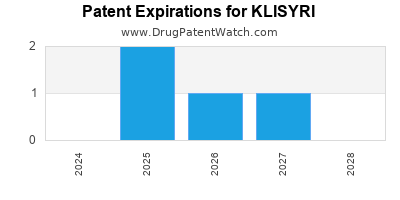

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for KLISYRI
Generic Entry Date for KLISYRI*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
OINTMENT;TOPICAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for KLISYRI
| Drug Class | Microtubule Inhibitor |
| Physiological Effect | Microtubule Inhibition |
Anatomical Therapeutic Chemical (ATC) Classes for KLISYRI
US Patents and Regulatory Information for KLISYRI
KLISYRI is protected by eight US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of KLISYRI is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting KLISYRI
Compositions for modulating a kinase cascade and methods of use thereof
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Methods of treating and/or preventing actinic keratosis
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TOPICAL TREATMENT OF ACTINIC KERATOSIS OF THE FACE OR SCALP
Solid forms of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TOPICAL TREATMENT OF ACTINIC KERATOSIS OF THE FACE OR SCALP
Compositions for treating cell proliferation disorders
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Composition and methods for modulating a kinase cascade
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TOPICAL TREATMENT OF ACTINIC KERATOSIS OF THE FACE OR SCALP
Biaryl compositions and methods for modulating a kinase cascade
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Compositions and methods of treating cell proliferation disorders
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting KLISYRI
NEW CHEMICAL ENTITY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for KLISYRI
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Almirall, S.A. | Klisyri | tirbanibulin | EMEA/H/C/005183 Klisyri is indicated for the field treatment of non-hyperkeratotic, non-hypertrophic actinic keratosis (Olsen grade 1) of the face or scalp in adults. |
Authorised | no | no | no | 2021-07-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for KLISYRI
When does loss-of-exclusivity occur for KLISYRI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 18330163
Estimated Expiration: ⤷ Try a Trial
Patent: 23201010
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 2020004419
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 74831
Estimated Expiration: ⤷ Try a Trial
China
Patent: 1278808
Patent: 2-(5-(4-(2-吗啉代乙氧基)苯基)吡啶-2-基)-N-苄基乙酰胺的固体形式 (SOLID FORMS OF 2-(5-(4-(2-MORPHOLINOETHOXY)PHENYL)PYRIDIN-2-YL)-N-BENZYLACETAMIDE)
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 79016
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 2992
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 20533412
Estimated Expiration: ⤷ Try a Trial
Patent: 23157909
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 20002622
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 20112539
Estimated Expiration: ⤷ Try a Trial
Singapore
Patent: 202001868R
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 200081359
Patent: 2--N-벤질아세트아미드의 고체 형태
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 15820
Estimated Expiration: ⤷ Try a Trial
Patent: 1920160
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering KLISYRI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2019051147 | ⤷ Try a Trial | |
| Japan | 2016222733 | キナーゼカスケードを調節するための組成物および方法 (COMPOSITIONS AND METHODS FOR MODULATING KINASE CASCADE) | ⤷ Try a Trial |
| Mexico | 2021013354 | METODOS PARA TRATAR Y/O PREVENIR QUERATOSIS ACTINICA. (METHODS OF TREATING AND/OR PREVENTING ACTINIC KERATOSIS.) | ⤷ Try a Trial |
| Denmark | 2041071 | ⤷ Try a Trial | |
| Australia | 2007265373 | Biaryl compositions and methods for modulating a kinase cascade | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for KLISYRI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1836169 | 49/2021 | Austria | ⤷ Try a Trial | PRODUCT NAME: TIRBANIBULIN ODER EIN SALZ, SOLVAT ODER HYDRAT DAVON; REGISTRATION NO/DATE: EU/1/21/1558 (MITTEILUNG) 20210719 |
| 1836169 | 21C1064 | France | ⤷ Try a Trial | PRODUCT NAME: TIRBANIBULINE; REGISTRATION NO/DATE: EU/1/21/1558 20210719 |
| 1836169 | C01836169/01 | Switzerland | ⤷ Try a Trial | FORMER OWNER: ATHENEX, INC., US |
| 1836169 | 122021000066 | Germany | ⤷ Try a Trial | PRODUCT NAME: KLISYRI TIRBANIBULIN; REGISTRATION NO/DATE: EU/1/21/1558 20210716 |
| 1836169 | 301145 | Netherlands | ⤷ Try a Trial | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), ASSIGNMENT |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


