EDURANT PED Drug Patent Profile
✉ Email this page to a colleague
When do Edurant Ped patents expire, and what generic alternatives are available?
Edurant Ped is a drug marketed by Janssen Prods and is included in one NDA.
The generic ingredient in EDURANT PED is rilpivirine hydrochloride. There are seven drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the rilpivirine hydrochloride profile page.
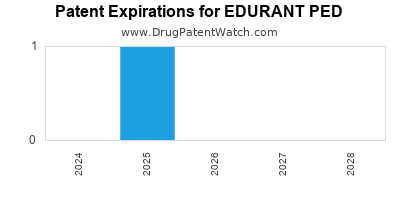
Summary for EDURANT PED
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 14 |
| Formulation / Manufacturing: | see details |
| DailyMed Link: | EDURANT PED at DailyMed |
Recent Clinical Trials for EDURANT PED
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Aptorum International Limited | Phase 1 |
| ViiV Healthcare | Phase 1/Phase 2 |
| National Institute of Allergy and Infectious Diseases (NIAID) | Phase 1/Phase 2 |
Pharmacology for EDURANT PED
Anatomical Therapeutic Chemical (ATC) Classes for EDURANT PED
US Patents and Regulatory Information for EDURANT PED
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janssen Prods | EDURANT PED | rilpivirine hydrochloride | TABLET, FOR SUSPENSION;ORAL | 219016-001 | Mar 15, 2024 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


