DIAZEPAM Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Diazepam, and what generic alternatives are available?
Diazepam is a drug marketed by Chartwell Molecular, Novel Labs Inc, Abraxis Pharm, Beloteca, Fresenius Kabi Usa, Hikma, Hospira, Marsam Pharms Llc, Parenta Pharms, Us Army, Warner Chilcott, Watson Labs, Watson Labs Inc, Actavis Elizabeth, Aurobindo Pharma Ltd, Barr, Chartwell Rx, Dava Pharms Inc, Dr Reddys Labs Sa, Duramed Pharms Barr, Ferndale Labs, Halsey, Ivax Sub Teva Pharms, Martec Usa Llc, Mylan, Nuvo Pharm, Pioneer Pharms, Roxane, Strides Pharma, Teva Pharms, and Virtus. and is included in seventy-six NDAs.
The generic ingredient in DIAZEPAM is diazepam. There are eight drug master file entries for this compound. Forty-two suppliers are listed for this compound. Additional details are available on the diazepam profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Diazepam
A generic version of DIAZEPAM was approved as diazepam by MYLAN on September 4th, 1985.
Summary for DIAZEPAM
| US Patents: | 0 |
| Applicants: | 31 |
| NDAs: | 76 |
| Finished Product Suppliers / Packagers: | 37 |
| Raw Ingredient (Bulk) Api Vendors: | 68 |
| Clinical Trials: | 138 |
| Patent Applications: | 465 |
| Formulation / Manufacturing: | see details |
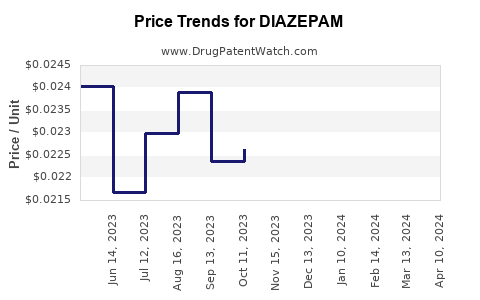
| Drug Prices: | Drug price information for DIAZEPAM |
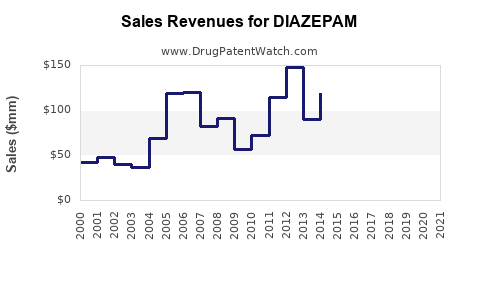
| Drug Sales Revenues: | Drug sales revenues for DIAZEPAM |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DIAZEPAM |
| What excipients (inactive ingredients) are in DIAZEPAM? | DIAZEPAM excipients list |
| DailyMed Link: | DIAZEPAM at DailyMed |
Recent Clinical Trials for DIAZEPAM
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Assiut University | N/A |
| Kuwait University | Early Phase 1 |
| Institute of Experimental and Clinical Therapeutics | Phase 2 |
Pharmacology for DIAZEPAM
| Drug Class | Benzodiazepine |
Medical Subject Heading (MeSH) Categories for DIAZEPAM
Anatomical Therapeutic Chemical (ATC) Classes for DIAZEPAM
Paragraph IV (Patent) Challenges for DIAZEPAM
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| DIASTAT ACUDIAL | Rectal Gel | diazepam | 5 mg/mL, 2mL pre-filled syringe | 020648 | 1 | 2008-12-23 |
| DIASTAT ACUDIAL | Rectal Gel | diazepam | 5 mg/mL, 4mL pre- filled syringe | 020648 | 1 | 2008-12-08 |
| DIASTAT ACUDIAL | Rectal Gel | diazepam | 2.5 mg/0.5 mL 5 mg/mL 10 mg/2 mL 15 mg/3 mL 20 mg/4 mL | 020648 | 1 | 2004-03-23 |
US Patents and Regulatory Information for DIAZEPAM
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Roxane | DIAZEPAM | diazepam | TABLET;ORAL | 070356-001 | Jun 17, 1986 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Chartwell Rx | DIAZEPAM | diazepam | TABLET;ORAL | 070303-001 | Dec 20, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Actavis Elizabeth | DIAZEPAM | diazepam | TABLET;ORAL | 070781-001 | Mar 19, 1986 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Watson Labs | DIAZEPAM | diazepam | TABLET;ORAL | 070458-001 | Nov 1, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Duramed Pharms Barr | DIAZEPAM | diazepam | TABLET;ORAL | 070894-001 | Aug 27, 1986 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ivax Sub Teva Pharms | DIAZEPAM | diazepam | TABLET;ORAL | 070361-001 | Sep 4, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


