Wyeth Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for WYETH PHARMS, and what generic alternatives to WYETH PHARMS drugs are available?
WYETH PHARMS has thirty-one approved drugs.
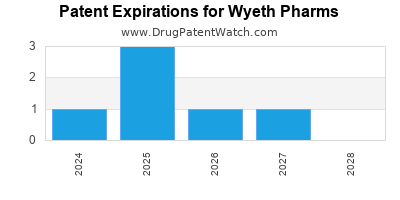
There are six US patents protecting WYETH PHARMS drugs.
There are fifty-six patent family members on WYETH PHARMS drugs in thirty countries and thirty-six supplementary protection certificates in thirteen countries.
Drugs and US Patents for Wyeth Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wyeth Pharms Inc | PIPRACIL | piperacillin sodium | INJECTABLE;INJECTION | 050545-002 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Wyeth Pharms Inc | LODINE XL | etodolac | TABLET, EXTENDED RELEASE;ORAL | 020584-001 | Oct 25, 1996 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Wyeth Pharms Inc | INDERIDE-40/25 | hydrochlorothiazide; propranolol hydrochloride | TABLET;ORAL | 018031-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Wyeth Pharms | INDERAL | propranolol hydrochloride | TABLET;ORAL | 016418-004 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Wyeth Pharms | INDERAL | propranolol hydrochloride | TABLET;ORAL | 016418-009 | Oct 18, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Wyeth Pharms | ZOSYN | piperacillin sodium; tazobactam sodium | INJECTABLE;INJECTION | 050684-002 | Oct 22, 1993 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Wyeth Pharms Inc | EFFEXOR | venlafaxine hydrochloride | TABLET;ORAL | 020151-005 | Dec 28, 1993 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Wyeth Pharms
Paragraph IV (Patent) Challenges for WYETH PHARMS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | For Injection | 12 g/1.5 g per vial (pharmacy bulk) | ➤ Subscribe | 2011-12-06 |
| ➤ Subscribe | Tablets | 0.09 mg/0.02 mg | ➤ Subscribe | 2007-10-05 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg and 40 mg | ➤ Subscribe | 2004-02-02 |
| ➤ Subscribe | Tablets | 25 mg, 37.5 mg, 50 mg, 75 mg and 100 mg | ➤ Subscribe | 2005-11-03 |
International Patents for Wyeth Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Singapore | 151331 | ⤷ Try a Trial |
| European Patent Office | 1732890 | ⤷ Try a Trial |
| Russian Federation | 2006132180 | ⤷ Try a Trial |
| Mexico | 266708 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2005100316 | ⤷ Try a Trial |
| South Korea | 20060118452 | ⤷ Try a Trial |
| China | 1886119 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Wyeth Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1539166 | 2013/055 | Ireland | ⤷ Try a Trial | PRODUCT NAME: THE COMBINATION OF: (A) DEXTROMETHORPHAN OR A PHARMACEUTICALLY ACCEPTABLE SALT, PRECURSOR OR DERIVATIVE THEREOF, E.G. DEXTROMETHORPHAN HYDROBROMIDE AND IN PARTICULAR DEXTROMETHORPHAN HYDROBROMIDE MONOHYDRATE; AND (B) QUINIDINE OR A PHARMACEUTICALLY ACCEPTABLE SALT, PRECURSOR OR DERIVATIVE THEREOF, E.G. QUINIDINE SULPHATE AND IN PARTICULAR QUINIDINE SULPHATE DIHYDRATE, PROTECTED BY THE BASIC PATENT; REGISTRATION NO/DATE: EU/1/13/833 20130624 |
| 1539166 | 13C0062 | France | ⤷ Try a Trial | PRODUCT NAME: (A) DEXTROMETHORPHAN OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE, PAR EXEMPLE LE BROMHYDRATE DE DEXTROMETHORPHAN ET EN PARTICULIER LE MONOHYDRATE DE BROMHYDRATE DE DEXTROMETHORPHAN (B) QUINIDINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE, PAR EXEMPLE LE SULFATE DE QUINIDINE EN PARTICULIER LE DIHYDRATE DE SULFATE DE QUINIDINE; REGISTRATION NO/DATE: EU/1/13/833/001-003 20130626 |
| 0503785 | CA 2011 00026 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF OLMESARTAN MEDOXOMIL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND AMLODIPINE BESYLATE AND HYDROCHLOROTHIAZIDE; NAT. REG. NO/DATE: 46260-46269 (DK) 20110323; FIRST REG. NO/DATE: DE 79810.00.00 20101216 |
| 0502314 | SPC/GB02/037 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: TELMISARTAN, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND HYDROCHLOROTHIAZIDE; REGISTERED: UK EU/1/02/213/001 20020419; UK EU/1/02/213/002 20020419; UK EU/1/02/213/003 20020419; UK EU/1/02/214/004 20020419; UK EU/1/02/213/005 20020419; UK EU/1/02/213/006 20020419; UK EU/1/02/213/007 20020419; UK EU/1/02/213/008 20020419; UK EU/1/02/213/009 20020419; UK EU/1/02/213/010 20020419 |
| 0443983 | C00443983/03 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN + AMLODIPINE + HYDROCHLOROTHIAZIDE; REGISTRATION NUMBER/DATE: SWISSMEDIC 59407 16.09.2009 |
| 1453521 | CA 2016 00016 | Denmark | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL OG ETHINYLOESTRADIOL; NAT. REG. NO/DATE: 56336 20151105; FIRST REG. NO/DATE: SK 17/0017/15-S 20150211 |
| 0802183 | 2009/028 | Ireland | ⤷ Try a Trial | PRODUCT NAME: BAZEDOXIFENE AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REGISTRATION NO/DATE: EU/1/09/511/001-004 20090417 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.