Viiv Hlthcare Company Profile
✉ Email this page to a colleague
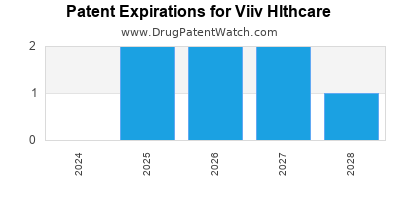
What is the competitive landscape for VIIV HLTHCARE, and when can generic versions of VIIV HLTHCARE drugs launch?
VIIV HLTHCARE has twenty-six approved drugs.
There are twelve US patents protecting VIIV HLTHCARE drugs.
There are five hundred and fourteen patent family members on VIIV HLTHCARE drugs in fifty-nine countries and two hundred and eleven supplementary protection certificates in eighteen countries.
Summary for Viiv Hlthcare
| International Patents: | 514 |
| US Patents: | 12 |
| Tradenames: | 19 |
| Ingredients: | 17 |
| NDAs: | 26 |
Drugs and US Patents for Viiv Hlthcare
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | RX | Yes | Yes | 11,224,597 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Viiv Hlthcare | TIVICAY PD | dolutegravir sodium | TABLET, FOR SUSPENSION;ORAL | 213983-001 | Jun 12, 2020 | RX | Yes | Yes | 8,129,385*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Viiv Hlthcare | DOVATO | dolutegravir sodium; lamivudine | TABLET;ORAL | 211994-001 | Apr 8, 2019 | RX | Yes | Yes | 9,242,986*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Viiv Hlthcare | TIVICAY | dolutegravir sodium | TABLET;ORAL | 204790-002 | Jun 9, 2016 | DISCN | Yes | No | 8,129,385*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-002 | Aug 6, 2007 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Viiv Hlthcare
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | RESCRIPTOR | delavirdine mesylate | TABLET;ORAL | 020705-001 | Apr 4, 1997 | 5,563,142 | ⤷ Try a Trial |
| Viiv Hlthcare | COMBIVIR | lamivudine; zidovudine | TABLET;ORAL | 020857-001 | Sep 26, 1997 | 6,180,639*PED | ⤷ Try a Trial |
| Viiv Hlthcare | JULUCA | dolutegravir sodium; rilpivirine hydrochloride | TABLET;ORAL | 210192-001 | Nov 21, 2017 | 8,101,629 | ⤷ Try a Trial |
| Viiv Hlthcare | ZIAGEN | abacavir sulfate | SOLUTION;ORAL | 020978-001 | Dec 17, 1998 | 6,294,540*PED | ⤷ Try a Trial |
| Viiv Hlthcare | ZIAGEN | abacavir sulfate | SOLUTION;ORAL | 020978-001 | Dec 17, 1998 | 5,089,500*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for VIIV HLTHCARE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 150 mg/300 mg | ➤ Subscribe | 2007-06-26 |
| ➤ Subscribe | Tablets | 10 mg, 25 mg and 50 mg | ➤ Subscribe | 2017-08-14 |
| ➤ Subscribe | Tablets | 300 mg/150 mg/300 mg | ➤ Subscribe | 2011-03-22 |
| ➤ Subscribe | Oral Solution | 10 mg/mL | ➤ Subscribe | 2011-11-22 |
| ➤ Subscribe | Tablets | 300 mg | ➤ Subscribe | 2009-01-28 |
| ➤ Subscribe | Tablets | 700 mg | ➤ Subscribe | 2012-01-18 |
| ➤ Subscribe | Tablets | 600 mg/300 mg | ➤ Subscribe | 2007-09-27 |
| ➤ Subscribe | Tablets | 600 mg/50 mg/300 mg | ➤ Subscribe | 2017-08-14 |
| ➤ Subscribe | Tablets | 150 mg and 300 mg | ➤ Subscribe | 2007-10-16 |
| ➤ Subscribe | Tablets | 150 mg and 300 mg | ➤ Subscribe | 2011-08-08 |
| ➤ Subscribe | Oral Solution | 20 mg/ml | ➤ Subscribe | 2012-12-27 |
International Patents for Viiv Hlthcare Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Croatia | P20080064 | ⤷ Try a Trial |
| Australia | 2019200813 | ⤷ Try a Trial |
| Australia | 2011302030 | ⤷ Try a Trial |
| Hungary | E038797 | ⤷ Try a Trial |
| Spain | 2384715 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Viiv Hlthcare Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1419152 | C20120012 00058 | Estonia | ⤷ Try a Trial | PRODUCT NAME: EVIPLERA - EMTRITSITABIIN/RILPIVIRIIN (HUEDRO-;REG NO/DATE: C(2011)8956 FINAL 28.11.2011 |
| 1284974 | C300338 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: MARAVIROC, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: EU/1/07/418/001-010 20070918 |
| 2465580 | PA2021512 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: KABOTEGRAVIRAS; REGISTRATION NO/DATE: EU/1/20/1481 20201217 |
| 3808743 | CA 2022 00035 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF RILPIVIRINE OR A THERAPEUTICALLY EQUIVALENT FORM THEREOF PROTECTED BY THE BASIC PATENT, SUCH AS A PHARMACEUTICALLY ACCEPTABLE ADDITION SALT OF RILPIVIRINE, INCLUDING THE HYDROCHLORIC ACID SALT OF RILPIVIRINE, AND EMTRICITABINE; REG. NO/DATE: EU/1/11/737/001-002 20111128 |
| 2932970 | C20180031 00270 | Estonia | ⤷ Try a Trial | PRODUCT NAME: DOLUTEGRAVIIR/RILPIVIRIIN;REG NO/DATE: EU/1/18/1282 18.05.2018 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.