Upjohn Company Profile
✉ Email this page to a colleague
What is the competitive landscape for UPJOHN, and when can generic versions of UPJOHN drugs launch?
UPJOHN has thirteen approved drugs.
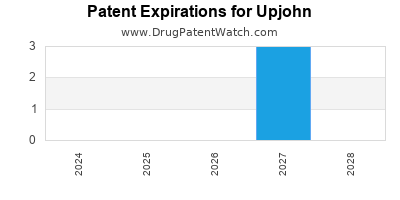
There are three US patents protecting UPJOHN drugs.
There are thirty-three patent family members on UPJOHN drugs in thirty-three countries and thirty-nine supplementary protection certificates in thirteen countries.
Drugs and US Patents for Upjohn
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Upjohn | EFFEXOR XR | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 020699-001 | Oct 20, 1997 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | EFFEXOR XR | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 020699-003 | Oct 20, 1997 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-005 | Dec 30, 2004 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | XANAX | alprazolam | TABLET;ORAL | 018276-004 | Nov 27, 1985 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | LYRICA CR | pregabalin | TABLET, EXTENDED RELEASE;ORAL | 209501-003 | Oct 11, 2017 | AB | RX | Yes | Yes | 8,945,620*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-001 | Sep 27, 2002 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | LYRICA CR | pregabalin | TABLET, EXTENDED RELEASE;ORAL | 209501-001 | Oct 11, 2017 | AB | RX | Yes | No | 9,144,559*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Upjohn
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-003 | Sep 27, 2002 | 6,863,902*PED | ⤷ Try a Trial |
| Upjohn | LIPITOR | atorvastatin calcium | TABLET;ORAL | 020702-004 | Apr 7, 2000 | 6,126,971*PED | ⤷ Try a Trial |
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | 7,163,959 | ⤷ Try a Trial |
| Upjohn | CELEBREX | celecoxib | CAPSULE;ORAL | 020998-001 | Dec 31, 1998 | RE44048*PED | ⤷ Try a Trial |
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-002 | Sep 27, 2002 | 6,558,707*PED | ⤷ Try a Trial |
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-002 | Sep 27, 2002 | 6,410,054*PED | ⤷ Try a Trial |
| Upjohn | CELEBREX | celecoxib | CAPSULE;ORAL | 020998-003 | Aug 29, 2002 | 5,760,068*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for UPJOHN drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Capsules | 2 mg and 4 mg | ➤ Subscribe | 2007-07-30 |
| ➤ Subscribe | Capsules | 50 mg | ➤ Subscribe | 2008-03-21 |
| ➤ Subscribe | Oral Solution | 20 mg/mL | ➤ Subscribe | 2010-05-19 |
| ➤ Subscribe | Extended-release Tablets | 82.5 mg and 165 mg | ➤ Subscribe | 2018-02-02 |
| ➤ Subscribe | Extended-release Tablets | 82.5 mg and 165 mg | ➤ Subscribe | 2018-02-02 |
| ➤ Subscribe | Tablets | 20 mg and 40 mg | ➤ Subscribe | 2010-03-29 |
| ➤ Subscribe | Extended-release Tablets | 37.5 mg, 75 mg and 150 mg | ➤ Subscribe | 2007-05-03 |
| ➤ Subscribe | Capsules | 25 mg, 50 mg, 75 mg, 100 mg, 150 mg, 200 mg, 225 mg and 300 mg | ➤ Subscribe | 2008-12-30 |
| ➤ Subscribe | Extended-release Tablets | 330 mg | ➤ Subscribe | 2018-01-29 |
| ➤ Subscribe | Extended-release Tablets | 330 mg | ➤ Subscribe | 2018-01-29 |
| ➤ Subscribe | Tablets | 25 mg and 50 mg | ➤ Subscribe | 2006-09-27 |
International Patents for Upjohn Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Eurasian Patent Organization | 012377 | ⤷ Try a Trial |
| Montenegro | 00482 | ⤷ Try a Trial |
| Portugal | 1945186 | ⤷ Try a Trial |
| Guatemala | 200600474 | ⤷ Try a Trial |
| Japan | 4334610 | ⤷ Try a Trial |
| Australia | 2006310217 | ⤷ Try a Trial |
| Norway | 20081816 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Upjohn Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0641330 | 2004C/022 | Belgium | ⤷ Try a Trial | PRODUCT NAME: PREGABALIN; REGISTRATION NO/DATE: EU/1/04/279/001 20040708 |
| 0641330 | 27/2004 | Austria | ⤷ Try a Trial | PRODUCT NAME: PREGABALIN, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES; REGISTRATION NO/DATE: EU/1/04/279/001 - EU/1/04/279/025 20040706 |
| 0364417 | C970039 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: [1R-[1 ALPHA (Z),2 BETA(R*),3 ALPHA,5ALPHA]]-7-[3,5-DIHYDROXY-2-(3-HYDROXY-5- FENYLPENTYL)CYCLOPENTYL]-5-HEPTEENZUUR, DESGEWENST IN DE VORM VAN EEN ZOUT OF EEN ESTER, IN HET BIJZONDER LATANOPROSTUM; NAT. REGISTRATION NO/DATE: RVG 21304 19970610; FIRST REGISTRATION: SE 12716 19960718 |
| 0592438 | 36/2001 | Austria | ⤷ Try a Trial | PRODUCT NAME: ELETRIPTAN UND DIE PHARMAZEUTISCH VERTRAEGLICHEN SALZE DAVON, EINSCHLIESSLICH DES HYDROBROMIDS; NAT. REGISTRATION NO/DATE: 1-24155 20010801; FIRST REGISTRATION: LI 55218 01, 55218 02 20001214 |
| 3461484 | 122021000036 | Germany | ⤷ Try a Trial | PRODUCT NAME: LATANOPROST, ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON, UND NETARSUDIL MESYLAT; REGISTRATION NO/DATE: EU/1/20/1502 20210107 |
| 0720599 | 92545 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: EZETIMIBE EN COMBINAISON AVEC ATORVASTATINE OU LEURS SELS PHARMACEUTIQUEMENT ACCEPTEES, Y COMPRIS ATORVASTATINE SOUS FORME D'ATORVASTATINE CALCIQUE TRIHYDRATEE; FIRST REGISTRATION: 20140910 |
| 0720599 | 300689 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: EZETIMIBE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, EN ATORVASTATINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER ATORVASTATINE CALCIUM TRIHYDRATE; NATIONAL REGISTRATION NO/DATE: RVG114373-114376 20141027; FIRST REGISTRATION: FR 2014091200122 20140912 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.