Salix Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SALIX, and what generic alternatives to SALIX drugs are available?
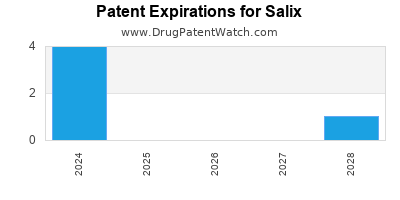
SALIX has eighteen approved drugs.
There are seventy-five US patents protecting SALIX drugs.
There are six hundred and forty-eight patent family members on SALIX drugs in fifty-five countries and ninety-seven supplementary protection certificates in twelve countries.
Summary for Salix
| International Patents: | 648 |
| US Patents: | 75 |
| Tradenames: | 15 |
| Ingredients: | 13 |
| NDAs: | 18 |
| Patent Litigation for Salix: | See patent lawsuits for Salix |
| PTAB Cases with Salix as patent owner: | See PTAB cases with Salix as patent owner |
Drugs and US Patents for Salix
Expired US Patents for Salix
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | 8,637,451 | ⤷ Try a Trial |
| Salix | UCERIS | budesonide | TABLET, EXTENDED RELEASE;ORAL | 203634-001 | Jan 14, 2013 | RE43799 | ⤷ Try a Trial |
| Salix | ZEGERID | omeprazole; sodium bicarbonate | FOR SUSPENSION;ORAL | 021636-002 | Dec 21, 2004 | 6,489,346 | ⤷ Try a Trial |
| Salix | ZEGERID | omeprazole; sodium bicarbonate | FOR SUSPENSION;ORAL | 021636-002 | Dec 21, 2004 | 5,840,737 | ⤷ Try a Trial |
| Salix | ZEGERID | omeprazole; sodium bicarbonate | CAPSULE;ORAL | 021849-001 | Feb 27, 2006 | 6,645,988 | ⤷ Try a Trial |
| Salix | APRISO | mesalamine | CAPSULE, EXTENDED RELEASE;ORAL | 022301-001 | Oct 31, 2008 | 8,956,647 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for SALIX drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Capsules | 0.375 g | ➤ Subscribe | 2012-04-03 |
| ➤ Subscribe | Powder for Oral Suspension | 40 mg/1680 mg per packet | ➤ Subscribe | 2007-08-24 |
| ➤ Subscribe | For Oral Solution | 140 g, 5.2 g, 2.2.g, 48.11 g, 9g and 7.54 g per pouch | ➤ Subscribe | 2018-12-06 |
| ➤ Subscribe | Injection | 12 mg/0.6 mL | ➤ Subscribe | 2015-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2016-09-06 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2010-08-24 |
| ➤ Subscribe | Tablets | 550 mg | ➤ Subscribe | 2015-12-18 |
| ➤ Subscribe | Extended-release Tablets | 9 mg | ➤ Subscribe | 2013-03-11 |
| ➤ Subscribe | Capsules | 20 mg/1100 mg and 40 mg/1100 mg | ➤ Subscribe | 2007-04-30 |
| ➤ Subscribe | Powder for Oral Suspension | 20mg/1680mg per packet | ➤ Subscribe | 2007-11-13 |
| ➤ Subscribe | Tablets | 40 mg and 120 mg | ➤ Subscribe | 2010-03-17 |
| ➤ Subscribe | Injection | 8 mg/0.4 mL | ➤ Subscribe | 2015-09-08 |
| ➤ Subscribe | For Oral Solution | 100 g, 7.5 g, 2.691 g, 1.015 g, 5.9 g and 4.7 g per pouch | ➤ Subscribe | 2007-11-27 |
| ➤ Subscribe | Tablets | 1.102 g and 0.398 g | ➤ Subscribe | 2008-04-09 |
| ➤ Subscribe | Tablets | 200 mg | ➤ Subscribe | 2019-01-28 |
International Patents for Salix Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Denmark | 2368553 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2005051361 | ⤷ Try a Trial |
| South Korea | 20060096242 | ⤷ Try a Trial |
| Hong Kong | 1092150 | ⤷ Try a Trial |
| Australia | 2018226473 | ⤷ Try a Trial |
| Hong Kong | 1199442 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Salix Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0788511 | SPC/GB08/036 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MICAFUNGIN AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF IN PARTICULAR MICAFUNGIN SODIUM; REGISTERED: UK EU/1/08/448/001 20080425; UK EU/1/08/448/002 20080425 |
| 0145340 | 99C0005 | Belgium | ⤷ Try a Trial | PRODUCT NAME: FOSPHENYTOIN DISODIUM; NAT. REGISTRATION NO/DATE: NL 23 613 19980806; FIRST REGISTRATION: GB - PL 000 19/0157 19980204 |
| 2203431 | 2015/009 | Ireland | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REGISTRATION NO/DATE: EU/1/14/983 20150115 |
| 0502314 | C300095 | Netherlands | ⤷ Try a Trial | PRODCUT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FYSIOLOGISCH VERDRAAGBAAR ZOUT, EN HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/02/213/001-010 20020419 |
| 2380576 | 2020/043 | Ireland | ⤷ Try a Trial | PRODUCT NAME: DEOXYCHOLIC ACID SODIUM SALT; NAT REGISTRATION NO/DATE: PA2103/003/001 20170602; FIRST REGISTRATION NO/DATE: SE/H/1547/001/DC 20160729 |
| 3141251 | 132021000000044 | Italy | ⤷ Try a Trial | PRODUCT NAME: UN PRODOTTO MEDICINALE COSTITUITO DA UNA COMBINAZIONE DI UNA PRIMA DOSE DI COMPOSIZIONE FARMACEUTICA E UNA SECONDA DOSE DI COMPOSIZIONE FARMACEUTICA, LA PRIMA DOSE DI COMPOSIZIONE FARMACEUTICA E COSTITUITA DAGLI INGREDIENTI ATTIVI POLIETILENGLICOLE, SOLFATO DI SODIO, CLORURO DI SODIO E CLORURO DI POTASSIO E LA SECONDA DOSE DI COMPOSIZIONE FARMACEUTICA E COSTITUITA DAGLI INGREDIENTI ATTIVI POLIETILENGLICOLE, ACIDO ASCORBICO, ASCORBATO DI SODIO, CLORURO DI SODIO E CLORURO DI POTASSIO.(PLENVU); AUTHORISATION NUMBER(S) AND DATE(S): IS/1/17/083/01, 20171016;045671017, 045671029, 045671031, 045671043, 045671056, 20171213 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.