Pharmacyclics Llc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PHARMACYCLICS LLC, and when can generic versions of PHARMACYCLICS LLC drugs launch?
PHARMACYCLICS LLC has three approved drugs.
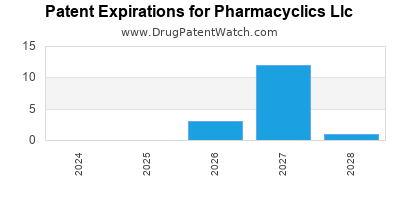
There are fifty-four US patents protecting PHARMACYCLICS LLC drugs.
There are three hundred and fifty patent family members on PHARMACYCLICS LLC drugs in forty-seven countries and thirty-four supplementary protection certificates in fifteen countries.
Summary for Pharmacyclics Llc
| International Patents: | 350 |
| US Patents: | 54 |
| Tradenames: | 1 |
| Ingredients: | 1 |
| NDAs: | 3 |
| Patent Litigation for Pharmacyclics Llc: | See patent lawsuits for Pharmacyclics Llc |
Drugs and US Patents for Pharmacyclics Llc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | TABLET;ORAL | 210563-003 | Feb 16, 2018 | RX | Yes | Yes | 8,952,015*PED | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | TABLET;ORAL | 210563-001 | Feb 16, 2018 | RX | Yes | No | 8,735,403*PED | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | TABLET;ORAL | 210563-003 | Feb 16, 2018 | RX | Yes | Yes | 9,801,881*PED | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | CAPSULE;ORAL | 205552-002 | Dec 20, 2017 | RX | Yes | No | 10,125,140*PED | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | CAPSULE;ORAL | 205552-002 | Dec 20, 2017 | RX | Yes | No | 10,961,251*PED | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | CAPSULE;ORAL | 205552-002 | Dec 20, 2017 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| Pharmacyclics Llc | IMBRUVICA | ibrutinib | SUSPENSION;ORAL | 217003-001 | Aug 24, 2022 | RX | Yes | Yes | 11,672,803 | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Paragraph IV (Patent) Challenges for PHARMACYCLICS LLC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Capsules | 70 mg | ➤ Subscribe | 2018-12-14 |
| ➤ Subscribe | Tablets | 280 mg and 420 mg | ➤ Subscribe | 2018-12-14 |
| ➤ Subscribe | Tablets | 140 mg and 560 mg | ➤ Subscribe | 2018-11-05 |
International Patents for Pharmacyclics Llc Drugs
Supplementary Protection Certificates for Pharmacyclics Llc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2529621 | 13/2017 | Austria | ⤷ Sign Up | PRODUCT NAME: IBRUTINIB ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/14/945/001-002 IDF DER FASSUNG DER ENTSCHEIDUNG DER KOMMISSION C(2015)4704 (MITTEILUNG) 20150707 |
| 2201840 | 233 5006-2015 | Slovakia | ⤷ Sign Up | PRODUCT NAME: IBRUTINIB; REGISTRATION NO/DATE: EU/1/14/945 20141023 |
| 2201840 | PA2015017,C2201840 | Lithuania | ⤷ Sign Up | PRODUCT NAME: IBRUTINIBAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/14/945 20141021 |
| 2526934 | 122016000093 | Germany | ⤷ Sign Up | PRODUCT NAME: LBRUTINIB ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/14/945 20160526 |
| 2529621 | 300868 | Netherlands | ⤷ Sign Up | PRODUCT NAME: IBRUTINIB, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: EU/1/14/945 - C(2015)4704 20150707 |
| 2201840 | C20150014 00145 | Estonia | ⤷ Sign Up | PRODUCT NAME: IBRUTINIIB;REG NO/DATE: EU/1/14/945 23.10.2014 |
| 2526934 | C20160038 00313 | Estonia | ⤷ Sign Up | PRODUCT NAME: IBRUTINIIB;REG NO/DATE: EU/1/14/945 30.05.2016 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.