Journey Company Profile
✉ Email this page to a colleague
What is the competitive landscape for JOURNEY, and what generic alternatives to JOURNEY drugs are available?
JOURNEY has eight approved drugs.
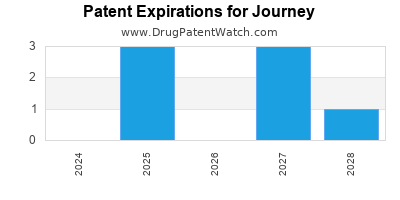
There are twenty-eight US patents protecting JOURNEY drugs.
There are seventy-one patent family members on JOURNEY drugs in seventeen countries and fifteen supplementary protection certificates in nine countries.
Summary for Journey
| International Patents: | 71 |
| US Patents: | 28 |
| Tradenames: | 6 |
| Ingredients: | 4 |
| NDAs: | 8 |
| Patent Litigation for Journey: | See patent lawsuits for Journey |
Drugs and US Patents for Journey
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Journey | AMZEEQ | minocycline hydrochloride | AEROSOL, FOAM;TOPICAL | 212379-001 | Oct 18, 2019 | RX | Yes | Yes | 10,398,641 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Journey | EURAX | crotamiton | CREAM;TOPICAL | 006927-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Journey | AMZEEQ | minocycline hydrochloride | AEROSOL, FOAM;TOPICAL | 212379-001 | Oct 18, 2019 | RX | Yes | Yes | 9,675,700 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Journey
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Journey | XIMINO | minocycline hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 201922-003 | Jul 11, 2012 | 5,908,838 | ⤷ Try a Trial |
| Journey | XIMINO | minocycline hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 201922-001 | Jul 11, 2012 | 5,908,838 | ⤷ Try a Trial |
| Journey | XIMINO | minocycline hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 201922-005 | Jul 11, 2012 | 5,908,838 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Journey Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Australia | 2016269524 | ⤷ Try a Trial |
| Australia | 2015224534 | ⤷ Try a Trial |
| New Zealand | 564093 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Journey Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1267866 | CA 2014 00020 | Denmark | ⤷ Try a Trial | PRODUCT NAME: GLYCOPYRRONIUM ELLER ET SALT DERAF, ISAER BROMIDSALTET, I KOMBINATION MED INDACATEROL ELLER ET SALT DERAF, ISAER MALEATSALTET; REG. NO/DATE: EU/1/13/862 20130919 |
| 1267866 | 92393 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: GLYCOPYRRONIUM OU UN SEL DE CELUI-CI, PLUS PARTICULIEREMENT LE SEL BROMURE, COMBINE A INDACATEROL OU UN SEL DE CELUI-CI, PLUS PARTICULIEREMENT LE SEL MALEATE. FIRST REGISTRATION: 20130923 |
| 1267866 | 2013/014 | Ireland | ⤷ Try a Trial | PRODUCT NAME: GLYCOPYRRONIUM OR A SALT THEREOF; REGISTRATION NO/DATE: EU/1/12/788/001-006 20120928 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.