Fresenius Medcl Company Profile
✉ Email this page to a colleague
What is the competitive landscape for FRESENIUS MEDCL, and when can generic versions of FRESENIUS MEDCL drugs launch?
FRESENIUS MEDCL has nine approved drugs.
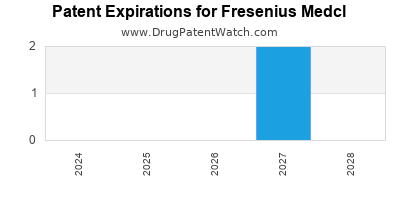
There are three US patents protecting FRESENIUS MEDCL drugs.
There are twenty-one patent family members on FRESENIUS MEDCL drugs in sixteen countries and ninety-three supplementary protection certificates in thirteen countries.
Summary for Fresenius Medcl
| International Patents: | 21 |
| US Patents: | 3 |
| Tradenames: | 22 |
| Ingredients: | 5 |
| NDAs: | 9 |
Drugs and US Patents for Fresenius Medcl
Expired US Patents for Fresenius Medcl
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Fresenius Medcl | PHOSLO GELCAPS | calcium acetate | CAPSULE;ORAL | 021160-003 | Apr 2, 2001 | 6,576,665 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO | calcium acetate | CAPSULE;ORAL | 021160-002 | Apr 2, 2001 | 6,576,665 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO | calcium acetate | CAPSULE;ORAL | 021160-001 | Apr 2, 2001 | 4,870,105 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO GELCAPS | calcium acetate | CAPSULE;ORAL | 021160-003 | Apr 2, 2001 | 4,870,105 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO GELCAPS | calcium acetate | CAPSULE;ORAL | 021160-003 | Apr 2, 2001 | 6,875,445 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO | calcium acetate | TABLET;ORAL | 019976-001 | Dec 10, 1990 | 4,870,105 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO | calcium acetate | CAPSULE;ORAL | 021160-002 | Apr 2, 2001 | 4,870,105 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for FRESENIUS MEDCL drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Capsules | EQ 169 mg calcium | ➤ Subscribe | 2005-05-31 |
| ➤ Subscribe | Oral Solution | 667 mg/5 mL | ➤ Subscribe | 2013-12-05 |
International Patents for Fresenius Medcl Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Canada | 2658465 | ⤷ Try a Trial |
| Japan | 5335674 | ⤷ Try a Trial |
| Spain | 2382993 | ⤷ Try a Trial |
| South Korea | 20090040340 | ⤷ Try a Trial |
| European Patent Office | 2048948 | ⤷ Try a Trial |
| Russian Federation | 2463060 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2008011126 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Fresenius Medcl Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2957286 | 2018/046 | Ireland | ⤷ Try a Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REGISTRATION NO/DATE: EU/1/17/1179 20170719 |
| 2666774 | LUC00167 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: RELEBACTAM, EVENTUELLEMENT SOUS FORME DE MONOHYDRATE, IMIPENEME ET CILASTATINE, EVENTUELLEMENT SOUS FORME DE SEL DE SODIUM; AUTHORISATION NUMBER AND DATE: EU/1/19/1420 20200217 |
| 2365988 | LUC00061 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM ET TOUS LES SELS ET DERIVES DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/17/1179 20170721 |
| 1912999 | 1490062-5 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SIMEPREVIR, OR A SALT THEREOF, INCLUDING SIMEPREVIR SODIUM; REG. NO/DATE: EU/1/14/924 20140516 |
| 3141251 | 301099 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: A MEDICINAL PRODUCT CONSISTING OF A COMBINATION OF A FIRST DOSE PHARMACEUTICAL COMPOSITION AND A SECOND DOSE PHARMACEUTICAL COMPOSITION, THE FIRST DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS POLYETHYLENE GLYCOL, SODIUM SULPHATE, SODIUM CHLORIDE AND POTASSIUM CHLORIDE AND THE SECOND DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS POLYETHYLENE GLYCOL, ASCORBIC ACID, SODIUM ASCORBATE, SODIUM CHLORIDE AND POTASSIUM CHLORIDE; NATIONAL REGISTRATION NO/DATE: RVG 120195 20171114; FIRST REGISTRATION: IS IS/1/17/083/01 20171016 |
| 2673237 | 1990016-6 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SODIUM ZIRCONIUM CYCLOSILICATE; REG. NO/DATE: EU/1/17/1173 20180326 |
| 0579826 | SPC/GB02/042 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ERTAPENEM ((1R,5S,6S,8R,2'S,4'S)-2-(2-(3-CARBOXYPHENYLCARBAMOYL)PYRROLIDIN-4-YLTHIO)-6-(1-HYDROXYETHYL)-1-METHYLCARBAPENEM-3-CARBOXYLIC ACID), A PHARMACEUTICALLY ACCEPTABLE SALT OR IN VIVO HYDROLYSABLE ESTER THEREOF, ESPECIALLY AS THE MONOSODIUM SALT ERTA; REGISTERED: UK EU/1/02/216/001 20020422 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.