Avion Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AVION PHARMS, and when can generic versions of AVION PHARMS drugs launch?
AVION PHARMS has three approved drugs.
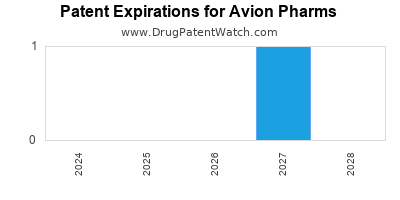
There are four US patents protecting AVION PHARMS drugs.
There are ten patent family members on AVION PHARMS drugs in seven countries and fifteen supplementary protection certificates in eight countries.
Drugs and US Patents for Avion Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Avion Pharms | PONSTEL | mefenamic acid | CAPSULE;ORAL | 015034-003 | Approved Prior to Jan 1, 1982 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Avion Pharms | DHIVY | carbidopa; levodopa | TABLET;ORAL | 214869-001 | Nov 12, 2021 | RX | Yes | Yes | 11,819,485 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Avion Pharms | DHIVY | carbidopa; levodopa | TABLET;ORAL | 214869-001 | Nov 12, 2021 | RX | Yes | Yes | 11,439,613 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Avion Pharms | DHIVY | carbidopa; levodopa | TABLET;ORAL | 214869-001 | Nov 12, 2021 | RX | Yes | Yes | 11,033,521 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Avion Pharms | BALCOLTRA | ethinyl estradiol; levonorgestrel | TABLET;ORAL | 208612-001 | Jan 9, 2018 | AB3 | RX | Yes | Yes | 7,838,042 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Avion Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Avion Pharms | BALCOLTRA | ethinyl estradiol; levonorgestrel | TABLET;ORAL | 208612-001 | Jan 9, 2018 | 6,716,814 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Avion Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 1737470 | ⤷ Try a Trial |
| Japan | 2022133449 | ⤷ Try a Trial |
| Brazil | 112020017422 | ⤷ Try a Trial |
| China | 111954523 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2005105112 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Avion Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1453521 | 39/2015 | Austria | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL UND EINE KOMBINATION VON LEVONORGESTREL UND ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: 136021 20150224; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 1453521 | 15C0050 | France | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL ET MELANGE DE LEVONORGESTREL ET ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: NL 42237 20150320; FIRST REGISTRATION: SK - 17/0017/15-S 20150129 |
| 0771217 | 07C0001 | France | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL BETADEX CLATHRATE; NAT. REGISTRATION NO/DATE: NL 32343 20060710; FIRST REGISTRATION: NL - RVG 31781 20050804 |
| 1380301 | 2009C/007 | Belgium | ⤷ Try a Trial | PRODUCT NAME: DROSPIRENONE-ETHINYLESTRADIOL; AUTHORISATION NUMBER AND DATE: BE321386 20080811 |
| 1214076 | SZ 49/2008 | Austria | ⤷ Try a Trial | PRODUCT NAME: WIRKSTOFFKOMBINATION VON ETHINYLESTRADIOL UND DROSPIRENON |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.