CABENUVA KIT Drug Patent Profile
✉ Email this page to a colleague
When do Cabenuva Kit patents expire, and what generic alternatives are available?
Cabenuva Kit is a drug marketed by Viiv Hlthcare and is included in one NDA. There are five patents protecting this drug.
This drug has three hundred and ninety-two patent family members in fifty-one countries.
The generic ingredient in CABENUVA KIT is cabotegravir; rilpivirine. One supplier is listed for this compound. Additional details are available on the cabotegravir; rilpivirine profile page.
DrugPatentWatch® Generic Entry Outlook for Cabenuva Kit
Cabenuva Kit will be eligible for patent challenges on January 21, 2025. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be September 15, 2031. This may change due to patent challenges or generic licensing.
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for CABENUVA KIT
| International Patents: | 392 |
| US Patents: | 5 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 2 |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CABENUVA KIT |
| DailyMed Link: | CABENUVA KIT at DailyMed |
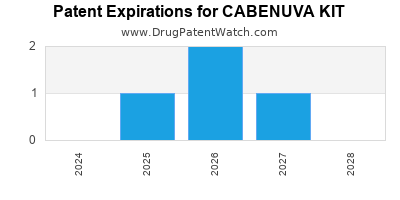
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for CABENUVA KIT
Generic Entry Date for CABENUVA KIT*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for CABENUVA KIT
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Allegheny Singer Research Institute (also known as Allegheny Health Network Research Institute) | Phase 3 |
| ViiV Healthcare | Phase 4 |
Anatomical Therapeutic Chemical (ATC) Classes for CABENUVA KIT
US Patents and Regulatory Information for CABENUVA KIT
CABENUVA KIT is protected by five US patents and three FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of CABENUVA KIT is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting CABENUVA KIT
N-[(2,4-difluorophenyl)methyl]-6-hydroxy-3-methyl-5,7-dioxo-2,3,5,7,11,11a- -hexahydro[1,3] oxazolo[3,2-a]pyrido[1,2-d]pyrazine-8-carboxamide having HIV integrase inhibitory activity
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HIV-1 INFECTION IN ADULTS AND ADOLESCENTS 12 YEARS OF AGE AND OLDER AND WEIGHING AT LEAST 35 KG
Aqueous suspensions of TMC278
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HIV-1 INFECTION IN ADULTS AND ADOLESCENTS 12 YEARS OF AGE AND OLDER AND WEIGHING AT LEAST 35 KG BY MONTHLY ADMINISTRATION OF RILPIVIRINE SUSPENSION AS PART OF COMBINATION THERAPY
HIV inhibiting pyrimidines derivatives
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HIV-1 INFECTION IN ADULTS AND ADOLESCENTS 12 YEARS OF AGE AND OLDER AND WEIGHING AT LEAST 35 KG
(3S,11aR)-N-[2,4-difluorophenyl)methyl]-6-hydroxy-3-methyl-5,7-dioxo-2,3,5- ,7,11,11a-hexahydro[1,3]oxazolo[3,2-a]pyrido[1,2-d]pyrazine-8-carboxamide useful as anti-HIV agent
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HIV-1 INFECTION IN ADULTS AND ADOLESCENTS 12 YEARS OF AGE AND OLDER AND WEIGHING AT LEAST 35 KG
FDA Regulatory Exclusivity protecting CABENUVA KIT
NEW CHEMICAL ENTITY
Exclusivity Expiration: ⤷ Try a Trial
NEW PATIENT POPULATION
Exclusivity Expiration: ⤷ Try a Trial
NEW DOSING SCHEDULE FOR CABOTEGRAVIR/RILPIVRINE INJECTION EVERY 2 MONTHS
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-002 | Jan 21, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for CABENUVA KIT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-002 | Jan 21, 2021 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-002 | Jan 21, 2021 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | CABENUVA KIT | cabotegravir; rilpivirine | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 212888-001 | Jan 21, 2021 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for CABENUVA KIT
When does loss-of-exclusivity occur for CABENUVA KIT?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 11302030
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 2013005907
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 10524
Estimated Expiration: ⤷ Try a Trial
Chile
Patent: 13000715
Estimated Expiration: ⤷ Try a Trial
China
Patent: 3547266
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0161280
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 18279
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 16076
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 4924
Estimated Expiration: ⤷ Try a Trial
Patent: 1390233
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 16076
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 31336
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 5028
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 31385
Estimated Expiration: ⤷ Try a Trial
Patent: 14500849
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 16076
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 6193
Estimated Expiration: ⤷ Try a Trial
Patent: 13003037
Estimated Expiration: ⤷ Try a Trial
Montenegro
Patent: 520
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 16076
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 16076
Estimated Expiration: ⤷ Try a Trial
San Marino
Patent: 01600350
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 222
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 16076
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 1301766
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1938662
Estimated Expiration: ⤷ Try a Trial
Patent: 130116254
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 94557
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 77377
Estimated Expiration: ⤷ Try a Trial
Patent: 1223529
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 8250
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering CABENUVA KIT around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Austria | 472537 | ⤷ Try a Trial | |
| Austria | 508748 | ⤷ Try a Trial | |
| Japan | WO2006088173 | HIVインテグラーゼ阻害活性を有する2環性カルバモイルピリドン誘導体 | ⤷ Try a Trial |
| Nicaragua | 200700068 | CLORHIDRATO DE 4-[[4-[[4-(2-CIANOETENIL)-2,6- DIMETILFENIL]AMINO]-2- PIRIMIDINIL]AMINO] BENZONITRILO | ⤷ Try a Trial |
| African Intellectual Property Organization (OAPI) | 12652 | HIV inhibiting pyrimidines derivatives. | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CABENUVA KIT
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1663240 | 2015/052 | Ireland | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF RILPIVIRINE OR A PHARMACEUTICALLY ACCEPTABLE SALT OF RILPIVIRINE, INCLUDING THE HYDROCHLORIDE SALT OF RILPIVIRINE, AND EMTRICITABINE; REGISTRATION NO/DATE: EU/1/11/737/001-002 20111128 |
| 1663240 | CA 2015 00055 | Denmark | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION AF RILPIVIRIN ELLER EN TERAPEUTISK AEKVIVALENT FORM DERAF, SOM ER BESKYTTET AF GRUNDPATENTET, SASOM ET FARMECEUTISK ACCEPTABELT SALT AF RILPIVIRIN, HERUNDER HYDROCHLORIDSALTET AF RILPIVIRIN, TENOFOVIR, NAVNLIG TENOFOVIRDISOPROXILFUMARAT; REG. NO/DATE: EU/1/11/737/001-002 20111128 |
| 1419152 | 628 | Finland | ⤷ Try a Trial | |
| 1632232 | CA 2016 00066 | Denmark | ⤷ Try a Trial | PRODUCT NAME: EN KOMBINATION AF: RILPIVIRINHYDROCHLORID ELLER EN TERAPEUTISK AEKVIVALENT FORM DERAF SOM ER BESKYTTET AF GRUNDPATENTET, EMTRICITABIN OG TENOFOVIRALAFENAMID, ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, SAERLIGT TENOFOVIRALAFENAMIDFUMARAT; REG. NO/DATE: EU/1/16/1112/001 20160623 |
| 1663240 | 2016/058 | Ireland | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF: RILPIVIRINE OR A THERAPEUTICALLY EQUIVALENT FORM THEREOF PROTECTED BY THE BASIC PATENT, SUCH AS A PHARMACEUTICALLY ACCEPTABLE SALT OF RILPIVIRINE, INCLUDING THE HYDROCHLORIDE SALT OF RILPIVIRINE; EMTRICITABINE; AND TENOFOVIR ALAFENAMIDE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR TENOFOVIR ALAFENAMIDE FUMARATE.; NAT REGISTRATION NO/DATE: EU/1/16/1112 20160621; FIRST REGISTRATION NO/DATE: JOURNAL OF THE INTELLECTUAL PROPERTY OFFICE OF IRELAND (NO. 2457) |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


