Teva Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TEVA PHARMS, and when can generic versions of TEVA PHARMS drugs launch?
TEVA PHARMS has three hundred and eighteen approved drugs.
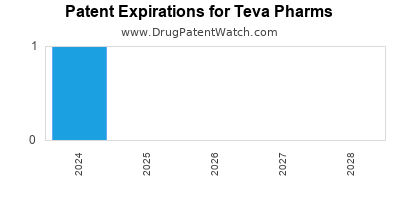
There are two US patents protecting TEVA PHARMS drugs. There are forty-four tentative approvals on TEVA PHARMS drugs.
There are seventeen patent family members on TEVA PHARMS drugs in twelve countries and nine hundred and sixteen supplementary protection certificates in eighteen countries.
Summary for Teva Pharms
| International Patents: | 17 |
| US Patents: | 2 |
| Tradenames: | 264 |
| Ingredients: | 259 |
| NDAs: | 318 |
Drugs and US Patents for Teva Pharms
Expired US Patents for Teva Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Teva Pharms Usa | COPAXONE | glatiramer acetate | INJECTABLE;SUBCUTANEOUS | 020622-002 | Feb 12, 2002 | 6,054,430 | ⤷ Try a Trial |
| Teva Pharms Usa | COPAXONE | glatiramer acetate | INJECTABLE;SUBCUTANEOUS | 020622-003 | Jan 28, 2014 | 8,367,605 | ⤷ Try a Trial |
| Teva Pharms Intl | AMRIX | cyclobenzaprine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 021777-001 | Feb 1, 2007 | 9,399,025 | ⤷ Try a Trial |
| Teva Pharms Usa | COPAXONE | glatiramer acetate | INJECTABLE;SUBCUTANEOUS | 020622-003 | Jan 28, 2014 | 6,054,430 | ⤷ Try a Trial |
| Teva Pharms Intl | AMRIX | cyclobenzaprine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 021777-001 | Feb 1, 2007 | 7,820,203 | ⤷ Try a Trial |
| Teva Pharms Usa | COPAXONE | glatiramer acetate | INJECTABLE;SUBCUTANEOUS | 020622-002 | Feb 12, 2002 | 6,342,476 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for TEVA PHARMS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Capsule | 15 mg and 30 mg | ➤ Subscribe | 2008-08-11 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
| ➤ Subscribe | Injection | 40 mg/mL, 1 mL pre-filled syringe | ➤ Subscribe | 2014-02-26 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | for Injection | 3.5 mg/vial | ➤ Subscribe | 2016-10-26 |
| ➤ Subscribe | Injection | 40 mg/mL, 1 mL pre-filled syringe | ➤ Subscribe | 2014-01-29 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
Premature patent expirations for TEVA PHARMS
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for Teva Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Australia | 2002337410 | ⤷ Try a Trial |
| Canada | 2459822 | ⤷ Try a Trial |
| Spain | 2383771 | ⤷ Try a Trial |
| Hong Kong | 1067562 | ⤷ Try a Trial |
| Austria | 451106 | ⤷ Try a Trial |
| Denmark | 2177223 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Teva Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1532149 | CA 2013 00001 | Denmark | ⤷ Try a Trial | PRODUCT NAME: 8-(3-AMINOPIPERIDIN-1-YL)-7-BUT-2-INYL-3-METHYL-1-(4-METHYLCHINAZOLIN-2-YLMETHYL)-3,7-DIHYDROPURIN-2,6-DION ENANTIOMERER OG SALTE DERAF - SAERLIGT LINAGLIPTIN - I KOMBINATION MED METFORMINHYDROCHLORID; REG. NO/DATE: EU/1/12/780/001-028 20120720 |
| 1663240 | 1690062-3 | Sweden | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF: RILPIVIRINE OR A PHARMACEUTICALLY ACCEPTABLE SALT OF RILPIVIRINE, INCLUDING THE HYDROCHLORIDE SALT OF RILPIVIRINE; EMTRICITABINE; AND TENOFOVIR ALAFENAMIDE, OR A PHARMCEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING TENOFOVIR ALAFENAMIDE FUMARATE.; REG. NO/DATE: EU/1/16/1112 20160623 |
| 0480717 | SPC/GB98/025 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MONTELUKAST, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, PREFERABLY MONTELUKAST SODIUM; REGISTERED: FI 12766 19970825; FI 12767 19970825; UK 00025/0357 19980115; UK 00025/0358 19980115 |
| 1869025 | 2018/034 | Ireland | ⤷ Try a Trial | PRODUCT NAME: BREXPIPRAZOLE OR A SALT THEREOF; NAT REGISTRATION NO/DATE: EU/1/18/1294 20180726; FIRST REGISTRATION NO/DATE: JOURNAL OF THE INTELLECTUAL PROPERTY OFFICE OF IRELAND (NO. 2402) |
| 0915894 | 05C0032 | France | ⤷ Try a Trial | PRODUCT NAME: TENOFOVIR DISOPROXIL FUMARATE; EMTRICITABINE; REGISTRATION NO/DATE: EU/1/04/305/001 20050221 |
| 0526171 | SPC/GB01/047 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: NATEGLINIDE; REGISTERED: CH 55401 01 20000928; CH 55401 02 20000928; UK EU/1/01/174/001 20010403; UK EU/1/01/174/002 20010403; UK EU/1/01/174/003 20010403; UK EU/1/01/174/004 20010403; UK EU/1/01/174/005 20010403; UK EU/1/01/174/006 20010403; UK EU/1/01/174/007 20010403; UK EU/1/01/174/008 20010403; UK EU/1/01/174/009 20010403; UK EU/1/01/174/010 20010403; UK EU/1/01/174/011 20010403; UK EU/1/01/174/012 20010403; UK EU/1/01/174/013 20010403; UK EU/1/01/174/014 20010403; UK EU/1/01/174/015 20010403; UK EU/1/01/174/016 20010403; UK EU/1/01/174/017 20010403; UK EU/1/01/174/018 20010403; UK EU/1/01/174/019 20010403; UK EU/1/01/174/020 20010403; UK EU/1/01/174/021 20010403 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.