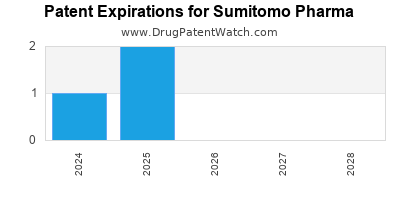
Sumitomo Pharma Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SUMITOMO PHARMA, and when can generic versions of SUMITOMO PHARMA drugs launch?
SUMITOMO PHARMA has four approved drugs.
There are forty-four US patents protecting SUMITOMO PHARMA drugs.
There are five hundred and thirty-six patent family members on SUMITOMO PHARMA drugs in forty-one countries and twenty-seven supplementary protection certificates in fifteen countries.
Summary for Sumitomo Pharma
| International Patents: | 536 |
| US Patents: | 44 |
| Tradenames: | 4 |
| Ingredients: | 4 |
| NDAs: | 4 |
Drugs and US Patents for Sumitomo Pharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-003 | May 21, 2020 | DISCN | Yes | No | 8,846,074 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-004 | May 21, 2020 | DISCN | Yes | No | 8,603,514 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | DISCN | Yes | No | 9,669,021 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-001 | Nov 8, 2013 | AB | RX | Yes | No | 9,566,244 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-004 | Nov 8, 2013 | AB | RX | Yes | Yes | 11,364,247 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | DISCN | Yes | No | 8,765,167 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Sumitomo Pharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-003 | May 21, 2020 | 8,765,167 | ⤷ Try a Trial |
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | 9,855,221 | ⤷ Try a Trial |
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-005 | May 21, 2020 | 9,931,305 | ⤷ Try a Trial |
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-004 | Nov 8, 2013 | 5,753,646 | ⤷ Try a Trial |
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-003 | Nov 8, 2013 | 5,753,646 | ⤷ Try a Trial |
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | 9,931,305 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Sumitomo Pharma Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 101346135 | ⤷ Try a Trial |
| China | 1768065 | ⤷ Try a Trial |
| Austria | E520665 | ⤷ Try a Trial |
| Japan | 2013501718 | ⤷ Try a Trial |
| Australia | 2019200308 | ⤷ Try a Trial |
| European Patent Office | 1463491 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Sumitomo Pharma Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1591446 | LUC00240 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: RELUGOLIX OU UN SEL DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/21/1565 20210720 |
| 1591446 | C 2021 047 | Romania | ⤷ Try a Trial | PRODUCT NAME: RELUGOLIX SAU O SARE A ACESTUIA; NATIONAL AUTHORISATION NUMBER: EU/1/21/1565; DATE OF NATIONAL AUTHORISATION: 20210716; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/21/1565; DATE OF FIRST AUTHORISATION IN EEA: 20210716 |
| 1591446 | C202130069 | Spain | ⤷ Try a Trial | PRODUCT NAME: RELUGOLIX O UNA SAL DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/21/1565; DATE OF AUTHORISATION: 20210716; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/21/1565; DATE OF FIRST AUTHORISATION IN EEA: 20210716 |
| 1591446 | CR 2021 00048 | Denmark | ⤷ Try a Trial | PRODUCT NAME: RELUGOLIX OR A SALT THEREOF; REG. NO/DATE: EU/1/21/1565 20210720 |
| 1591446 | 21C1056 | France | ⤷ Try a Trial | PRODUCT NAME: RELUGOLIX OU UN SEL DE CELUI-CI; REGISTRATION NO/DATE: EU/1/21/1565 20210720 |
| 1591446 | 2190051-9 | Sweden | ⤷ Try a Trial | PRODUCT NAME: RELUGOLIX OR A SALT THEREOF; REG. NO/DATE: EU/1/21/1565 20210720 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.