Pfizer Company Profile
✉ Email this page to a colleague
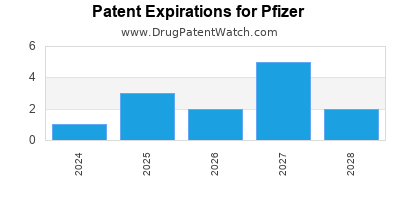
What is the competitive landscape for PFIZER, and when can generic versions of PFIZER drugs launch?
PFIZER has one hundred and ninety-five approved drugs.
There are forty-nine US patents protecting PFIZER drugs. There is one tentative approval on PFIZER drugs.
There are one thousand and twenty-four patent family members on PFIZER drugs in seventy countries and two hundred and sixty supplementary protection certificates in nineteen countries.
Summary for Pfizer
| International Patents: | 1024 |
| US Patents: | 49 |
| Tradenames: | 159 |
| Ingredients: | 130 |
| NDAs: | 195 |
| Drug Master File Entries: | 3 |
| Patent Litigation for Pfizer: | See patent lawsuits for Pfizer |
| PTAB Cases with Pfizer as petitioner: | See PTAB cases with Pfizer as petitioner |
Drugs and US Patents for Pfizer
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pfizer | CAVERJECT | alprostadil | INJECTABLE;INJECTION | 020379-001 | Jul 6, 1995 | AP | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Pfizer | CALAN | verapamil hydrochloride | TABLET;ORAL | 018817-003 | Feb 23, 1988 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pfizer | TROVAN PRESERVATIVE FREE | alatrofloxacin mesylate | INJECTABLE;INJECTION | 020760-002 | Dec 18, 1997 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pfizer | ATARAX | hydroxyzine hydrochloride | TABLET;ORAL | 010392-005 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pfizer | TOVIAZ | fesoterodine fumarate | TABLET, EXTENDED RELEASE;ORAL | 022030-002 | Oct 31, 2008 | AB | RX | Yes | Yes | 7,807,715*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Pfizer | FLAGYL I.V. | metronidazole hydrochloride | INJECTABLE;INJECTION | 018353-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Pfizer
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pfizer | TROVAN | trovafloxacin mesylate | TABLET;ORAL | 020759-001 | Dec 18, 1997 | 5,164,402 | ⤷ Try a Trial |
| Pfizer | COVERA-HS | verapamil hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 020552-001 | Feb 26, 1996 | 5,200,196 | ⤷ Try a Trial |
| Pfizer Pharms | ACCURETIC | hydrochlorothiazide; quinapril hydrochloride | TABLET;ORAL | 020125-003 | Dec 28, 1999 | 4,344,949*PED | ⤷ Try a Trial |
| Pfizer | ZAVZPRET | zavegepant hydrochloride | SPRAY, METERED;NASAL | 216386-001 | Mar 9, 2023 | 7,314,883 | ⤷ Try a Trial |
| Pfizer | DEPO-PROVERA | medroxyprogesterone acetate | INJECTABLE;INJECTION | 012541-002 | Approved Prior to Jan 1, 1982 | 4,038,389 | ⤷ Try a Trial |
| Pfizer | PROCARDIA XL | nifedipine | TABLET, EXTENDED RELEASE;ORAL | 019684-002 | Sep 6, 1989 | 5,264,446 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for PFIZER drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 1 g | ➤ Subscribe | 2005-08-23 |
| ➤ Subscribe | Delayed-release Tablets | 75 mg/0.2 mg | ➤ Subscribe | 2008-11-28 |
| ➤ Subscribe | Extended-release Tablets | 4 mg and 8 mg | ➤ Subscribe | 2012-10-31 |
| ➤ Subscribe | Injection | 20 mg/mL, 2 mL and 5 mL vials | ➤ Subscribe | 2004-07-26 |
| ➤ Subscribe | Capsules | 5 mg and 10 mg | ➤ Subscribe | 2005-06-21 |
| ➤ Subscribe | For Injection | 500 mg/vial | ➤ Subscribe | 2011-06-17 |
| ➤ Subscribe | Oral Suspension | 100 mg/5 mL | ➤ Subscribe | 2009-08-03 |
| ➤ Subscribe | Injection | 2 mg/mL, 100 mL bag | ➤ Subscribe | 2009-12-29 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Delayed-release Tablets | 50 mg/0.2 mg | ➤ Subscribe | 2009-06-29 |
| ➤ Subscribe | Extended-release Tablets | 11 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Capsules | 20 mg, 40 mg, 60 mg and 80 mg | ➤ Subscribe | 2005-02-07 |
| ➤ Subscribe | Capsules | 75 mg, 100 mg and 125 mg | ➤ Subscribe | 2019-02-04 |
| ➤ Subscribe | Capsules | 0.125 mg, 0.25 mg, and 0.5 mg | ➤ Subscribe | 2014-05-01 |
| ➤ Subscribe | Tablets | 600 mg | ➤ Subscribe | 2005-12-21 |
| ➤ Subscribe | Injection | 2 mg/mL, 300 mL bag | ➤ Subscribe | 2009-09-01 |
International Patents for Pfizer Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 108743588 | ⤷ Try a Trial |
| Lithuania | PA2022516 | ⤷ Try a Trial |
| Japan | 2014162794 | ⤷ Try a Trial |
| China | 111419817 | ⤷ Try a Trial |
| Hungary | E054212 | ⤷ Try a Trial |
| Eurasian Patent Organization | 200971104 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Pfizer Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2170860 | LUC00173 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: GLASDEGIB, EVENTUELLEMENT SOUS LA FORME D'UN SEL PHARMACEUTIQUEMENT ACCEPTABLE, Y COMPRIS LE SEL DE MALEATE; AUTHORISATION NUMBER AND DATE: EU/1/20/1451 20200629 |
| 2958921 | PA2022502 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: ABROCITINIBAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; REGISTRATION NO/DATE: EU/1/21/1593 20211209 |
| 1667986 | 92172 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: SOLVAT ACETONIQUE DU CABAZITAXEL, OU DESIGNE SOLVAT ACETONIQUE DU DIMETHOXY DOCETAXEL OU SOLVAT ACETONIQUE DU (2R,3S)-3-TERT-BUTOXYCARBONYLAMINO-2-HYDROXY-3-PHENYLPROPIONATE DE 4-ACETOXY-2A-BENZOYLOXY-5BETA,20-EPOXY-1-HYDROXY-7BETA,10A-DIMETHOXY-9-OXO-TAX-11-ENE-13A-YLE(ACETONATE DU CABAZITAXEL) |
| 1453521 | 15C0050 | France | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL ET MELANGE DE LEVONORGESTREL ET ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: NL 42237 20150320; FIRST REGISTRATION: SK - 17/0017/15-S 20150129 |
| 0136011 | 99C0003 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL AND NORETHISTERONE; FIRST REGISTRATION NO/DATE: 403 IS 106 F3 19980928; FIRST REGISTRATION: SE 14007 19980306 |
| 0770388 | SPC/GB09/026 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL AND COMBINATIONS OF ESTRADIOL AND DIENOGEST, PREFERABLY ESTRADIOL VALERATE AND COMBINATIONS OF ESTRADIOL VALERATE AND DIENOGEST; REGISTERED: BE BE 327792 20081103; UK PL 00010/0576-0001 20081208 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.