Harrow Eye Company Profile
✉ Email this page to a colleague
What is the competitive landscape for HARROW EYE, and what generic alternatives to HARROW EYE drugs are available?
HARROW EYE has nineteen approved drugs.
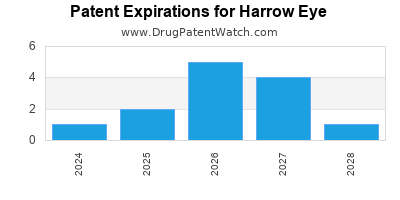
There are twenty-nine US patents protecting HARROW EYE drugs.
There are three hundred and fifty-four patent family members on HARROW EYE drugs in forty-two countries and twenty-five supplementary protection certificates in ten countries.
Drugs and US Patents for Harrow Eye
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Harrow Eye | VERKAZIA | cyclosporine | EMULSION;OPHTHALMIC | 214965-001 | Jun 23, 2021 | RX | Yes | Yes | 9,132,071 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Harrow Eye | VERKAZIA | cyclosporine | EMULSION;OPHTHALMIC | 214965-001 | Jun 23, 2021 | RX | Yes | Yes | 9,220,694 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | DISCN | Yes | No | 8,450,311 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Harrow Eye | VERKAZIA | cyclosporine | EMULSION;OPHTHALMIC | 214965-001 | Jun 23, 2021 | RX | Yes | Yes | 9,956,289 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Harrow Eye | VIGAMOX | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 021598-001 | Apr 15, 2003 | AT1 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Harrow Eye
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | 4,990,517*PED | ⤷ Try a Trial |
| Harrow Eye | TOBRASONE | fluorometholone acetate; tobramycin | SUSPENSION/DROPS;OPHTHALMIC | 050628-001 | Jul 21, 1989 | 5,149,693 | ⤷ Try a Trial |
| Harrow Eye | NEVANAC | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 021862-001 | Aug 19, 2005 | 5,475,034 | ⤷ Try a Trial |
| Harrow Eye | ILEVRO | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 203491-001 | Oct 16, 2012 | 6,403,609 | ⤷ Try a Trial |
| Harrow Eye | TRIESENCE | triamcinolone acetonide | INJECTABLE;INTRAVITREAL | 022048-001 | Nov 29, 2007 | 6,395,294 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for HARROW EYE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Ophthalmic Solution | 10.5% | ➤ Subscribe | 2012-02-29 |
| ➤ Subscribe | Ophthalmic Suspension | 0.3% | ➤ Subscribe | 2015-12-21 |
| ➤ Subscribe | Ophthalmic | 0.50% | ➤ Subscribe | 2005-12-22 |
International Patents for Harrow Eye Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Spain | 2910374 | ⤷ Try a Trial |
| Portugal | 2262506 | ⤷ Try a Trial |
| Canada | 2781254 | ⤷ Try a Trial |
| South Korea | 20120062846 | ⤷ Try a Trial |
| Japan | 6033677 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Harrow Eye Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0350733 | 11/2000 | Austria | ⤷ Try a Trial | PRODUCT NAME: MOXIFLOXACIN, DESSEN PHARMAZEUTISCH VERWENDBARE HYDRATE UND SAEUREADDITIONSSALZE, DESSEN ALKALI-ERDALKALI-SILBER- UND GUANIDINIUMSALZE, SOWIE DESSEN C1 - C4 ALKYL- ODER; NAT. REGISTRATION NO/DATE: 1-23494, 1-23495, 1-23496 20000215; FIRST REGISTRATION: DE 45263.00.00 19990621 |
| 0350733 | C300111 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: MOXIFLOXACINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER MOXIFLOXACINEHYDROCHLORIDE; NAT. REGISTRATION NO/DATE: RVG 28118 RVG 28119 20021017; FIRST REGISTRATION: DE 45263.00.00 19990621 |
| 2049079 | LUC00006 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: CYCLOSPORINE (GOUTTES OCULAIRES SOUS FORME D'EMULSION); AUTHORISATION NUMBER AND DATE: EU/1/15/990 20150323 |
| 0058146 | 2001C/045 | Belgium | ⤷ Try a Trial | PRODUCT NAME: DICHLORHYDRATE DE LEVOCETIRIZINE; NAT RER. NO/DATE: 194 IS 90 F3 20011022; FIRST REG.: DE 49903.00.00 20010103 |
| 2049079 | PA2017003,C2049079 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: CIKLOSPORINAS; REGISTRATION NO/DATE: EU/1/15/990 20170319 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.