Genentech Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GENENTECH INC, and when can generic versions of GENENTECH INC drugs launch?
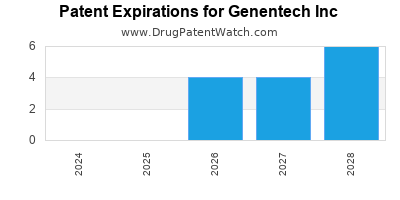
GENENTECH INC has eight approved drugs.
There are fifty-four US patents protecting GENENTECH INC drugs.
There are eight hundred and eighteen patent family members on GENENTECH INC drugs in fifty-three countries and sixty-seven supplementary protection certificates in nineteen countries.
Summary for Genentech Inc
| International Patents: | 818 |
| US Patents: | 54 |
| Tradenames: | 5 |
| Ingredients: | 5 |
| NDAs: | 8 |
| Drug Master File Entries: | 7 |
| Patent Litigation for Genentech Inc: | See patent lawsuits for Genentech Inc |
Drugs and US Patents for Genentech Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genentech Inc | ESBRIET | pirfenidone | TABLET;ORAL | 208780-003 | Jan 11, 2017 | AB | RX | Yes | Yes | 7,910,610 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Genentech Inc | ROZLYTREK | entrectinib | CAPSULE;ORAL | 212725-002 | Aug 15, 2019 | RX | Yes | Yes | 10,561,651 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Genentech Inc | ESBRIET | pirfenidone | CAPSULE;ORAL | 022535-001 | Oct 15, 2014 | AB | RX | Yes | Yes | 8,778,947 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Genentech Inc | ESBRIET | pirfenidone | CAPSULE;ORAL | 022535-001 | Oct 15, 2014 | AB | RX | Yes | Yes | 7,767,225 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Genentech Inc | ESBRIET | pirfenidone | CAPSULE;ORAL | 022535-001 | Oct 15, 2014 | AB | RX | Yes | Yes | 7,696,236 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Genentech Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Genentech Inc | ESBRIET | pirfenidone | TABLET;ORAL | 208780-001 | Jan 11, 2017 | 9,561,217 | ⤷ Try a Trial |
| Genentech Inc | ESBRIET | pirfenidone | TABLET;ORAL | 208780-003 | Jan 11, 2017 | 9,561,217 | ⤷ Try a Trial |
| Genentech Inc | ESBRIET | pirfenidone | TABLET;ORAL | 208780-002 | Jan 11, 2017 | 9,561,217 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for GENENTECH INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 267 mg, 534 mg and 801 mg | ➤ Subscribe | 2018-10-15 |
International Patents for Genentech Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 6914202 | ⤷ Try a Trial |
| China | 101365676 | ⤷ Try a Trial |
| European Patent Office | 3498281 | ⤷ Try a Trial |
| European Patent Office | 3143025 | ⤷ Try a Trial |
| China | 110494141 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Genentech Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2176231 | 20C1064 | France | ⤷ Try a Trial | PRODUCT NAME: ENTRECTINIB OU SES TAUTOMERES OU SELS PHARMACEUTIQUEMENT ACCEPTABLES; REGISTRATION NO/DATE: EU/1/20/1460 20200803 |
| 1934174 | PA2016016 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: KOBIMETINIBAS; REGISTRATION NO/DATE: EU/1/15/1048 20151120 |
| 3143025 | 132021000000149 | Italy | ⤷ Try a Trial | PRODUCT NAME: RISDIPLAM O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE(EVRYSDI); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/21/1531, 20210329 |
| 1934174 | 93078 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COBIMETINIB, EVENTUELLEMENT SOUS TOUTE FORME PROTEGEE PAR LE BREVET DE BASE, Y COMPRIS LES SELS ET SOLVATES PHARMACEUTIQUEMENT ACCEPTABLES, EN PARTICULIER HEMIFUMARATE DE COBIMETINIB; FIRST REGISTRATION DATE: 20151124 |
| 2176231 | 132020000000164 | Italy | ⤷ Try a Trial | PRODUCT NAME: ENTRECTINIB O SUOI ISOMERI, TAUTOMERI O SALI FARMACEUTICAMENTE ACCETTABILI(ROZLYTREK); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/20/1460, 20200803 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.