Fresenius Medcl Company Profile
✉ Email this page to a colleague
What is the competitive landscape for FRESENIUS MEDCL, and when can generic versions of FRESENIUS MEDCL drugs launch?
FRESENIUS MEDCL has nine approved drugs.
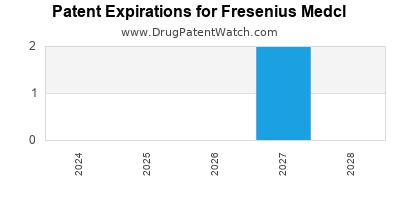
There are three US patents protecting FRESENIUS MEDCL drugs.
There are twenty-one patent family members on FRESENIUS MEDCL drugs in sixteen countries and ninety-three supplementary protection certificates in thirteen countries.
Summary for Fresenius Medcl
| International Patents: | 21 |
| US Patents: | 3 |
| Tradenames: | 22 |
| Ingredients: | 5 |
| NDAs: | 9 |
Drugs and US Patents for Fresenius Medcl
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresenius Medcl | PHOSLO GELCAPS | calcium acetate | CAPSULE;ORAL | 021160-003 | Apr 2, 2001 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Fresenius Medcl | CALCITRIOL | calcitriol | INJECTABLE;INJECTION | 075766-001 | Feb 20, 2003 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Fresenius Medcl | DELFLEX W/ DEXTROSE 4.25% LOW MAGNESIUM IN PLASTIC CONTAINER | calcium chloride; dextrose; magnesium chloride; sodium chloride; sodium lactate | SOLUTION;INTRAPERITONEAL | 018883-006 | Nov 30, 1984 | AT | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Fresenius Medcl
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Fresenius Medcl | PHOSLO | calcium acetate | TABLET;ORAL | 019976-001 | Dec 10, 1990 | 4,870,105 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO | calcium acetate | CAPSULE;ORAL | 021160-002 | Apr 2, 2001 | 6,576,665 | ⤷ Try a Trial |
| Fresenius Medcl | PHOSLO | calcium acetate | CAPSULE;ORAL | 021160-001 | Apr 2, 2001 | 4,870,105 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for FRESENIUS MEDCL drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Oral Solution | 667 mg/5 mL | ➤ Subscribe | 2013-12-05 |
| ➤ Subscribe | Capsules | EQ 169 mg calcium | ➤ Subscribe | 2005-05-31 |
International Patents for Fresenius Medcl Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Hong Kong | 1202815 | ⤷ Try a Trial |
| Brazil | PI0714882 | ⤷ Try a Trial |
| Japan | 2009544615 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Fresenius Medcl Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2957286 | LUC00094 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; AUTHORISATION NUMBER AND DATE: EU/1/17/1179 20170721 |
| 1713823 | 1490064-1 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SIMEPREVIR, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING SIMEPREVIR SODIUM; REG. NO/DATE: EU/1/14/924 20140516 |
| 2365988 | LUC00061 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM ET TOUS LES SELS ET DERIVES DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/17/1179 20170721 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.