Cheplapharm Company Profile
✉ Email this page to a colleague
What is the competitive landscape for CHEPLAPHARM, and when can generic versions of CHEPLAPHARM drugs launch?
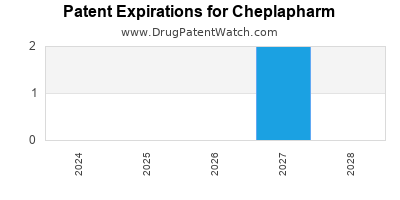
CHEPLAPHARM has thirteen approved drugs.
There are two US patents protecting CHEPLAPHARM drugs.
There are thirty-two patent family members on CHEPLAPHARM drugs in thirty-one countries and twenty-eight supplementary protection certificates in thirteen countries.
Drugs and US Patents for Cheplapharm
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cheplapharm | VALCYTE | valganciclovir hydrochloride | FOR SOLUTION;ORAL | 022257-001 | Aug 28, 2009 | AB | RX | Yes | Yes | 9,642,911 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Cheplapharm | ZYPREXA RELPREVV | olanzapine pamoate | SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 022173-003 | Dec 11, 2009 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Cheplapharm | KLONOPIN | clonazepam | TABLET;ORAL | 017533-001 | Approved Prior to Jan 1, 1982 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Cheplapharm | XENICAL | orlistat | CAPSULE;ORAL | 020766-001 | Apr 23, 1999 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Cheplapharm | ETOPOPHOS PRESERVATIVE FREE | etoposide phosphate | INJECTABLE;INJECTION | 020457-001 | May 17, 1996 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Cheplapharm
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Cheplapharm | ZYPREXA | olanzapine | TABLET;ORAL | 020592-002 | Sep 30, 1996 | 5,919,485*PED | ⤷ Try a Trial |
| Cheplapharm | ZYPREXA | olanzapine | TABLET;ORAL | 020592-005 | Sep 9, 1997 | 5,627,178*PED | ⤷ Try a Trial |
| Cheplapharm | ETOPOPHOS PRESERVATIVE FREE | etoposide phosphate | INJECTABLE;INJECTION | 020457-001 | May 17, 1996 | 5,041,424 | ⤷ Try a Trial |
| Cheplapharm | VALCYTE | valganciclovir hydrochloride | TABLET;ORAL | 021304-001 | Mar 29, 2001 | 6,083,953*PED | ⤷ Try a Trial |
| Cheplapharm | ZYPREXA | olanzapine | TABLET;ORAL | 020592-006 | Sep 9, 1997 | 5,736,541*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for CHEPLAPHARM drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 450 mg | ➤ Subscribe | 2005-12-27 |
| ➤ Subscribe | Tablets | 150 mg and 500 mg | ➤ Subscribe | 2008-11-10 |
| ➤ Subscribe | For Oral Solution | 50 mg/mL | ➤ Subscribe | 2011-03-21 |
International Patents for Cheplapharm Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Canada | 2671470 | ⤷ Try a Trial |
| Portugal | 2101733 | ⤷ Try a Trial |
| Russian Federation | 2009126616 | ⤷ Try a Trial |
| Cyprus | 1113468 | ⤷ Try a Trial |
| Japan | 2010513237 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Cheplapharm Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0454436 | SPC/GB96/058 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: OLANZAPINE OPTIONALLY IN THE FORM OF AN ACID ADDITION SALT; REGISTERED: UK EU/1/96/022/001 19960927; UK EU/1/96/022/002 19960927; UK EU/1/96/022/003 19960927; UK EU/1/96/022/004 19960927; UK EU/1/96/022/005 19960927; UK EU/1/96/022/006 19960927; UK EU/1/96/022/007 19960927; UK EU/1/96/022/008 19960927; UK EU/1/96/022/009 19960927; UK EU/1/96/022/010 19960927 |
| 1304992 | 92401 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: CLINDAMYCINE(EN TANT QUE PHOPSHATE DE CLINDAMYCINE)ET TRETINOINE |
| 0694547 | 2002/028 | Ireland | ⤷ Try a Trial | PRODUCT NAME: VALGANCICLOVIR (2-(2-AMINO-1,6-DIHYDRO-6-OXO-PURIN-9-YL)- METHOXY-3-HYDROXY-1-PROPANYL-L-VALINATE) AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; NAT REGISTRATION NO/DATE: 50/150/1 20020913; FIRST REGISTRATION NO/DATE: NL 25992 20010920; PAEDIATRIC INVESTIGATION PLAN: P/0220/2013 |
| 0454436 | C970015 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: OLANZAPINE, DESGEWENST IN DE VORM VAN EEN ZUURADDITIEZOUT; REGISTRATION NO/DATE: EU/1/96/022/001 - EU/1/96/022/010 19960927 |
| 0617614 | C300043 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: ALITRETINOIN; NAT. REGISTRATION NO/DATE: EU/1/00/149/001 20001011; FIRST REGISTRATION: EU/1/00/149/001 20001011 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.