Bionpharma Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BIONPHARMA, and when can generic versions of BIONPHARMA drugs launch?
BIONPHARMA has sixty-six approved drugs.
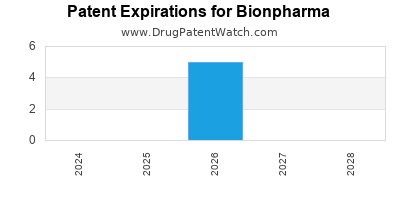
There are five US patents protecting BIONPHARMA drugs.
There are ten patent family members on BIONPHARMA drugs in thirteen countries and one hundred and seven supplementary protection certificates in fifteen countries.
Summary for Bionpharma
| International Patents: | 10 |
| US Patents: | 5 |
| Tradenames: | 61 |
| Ingredients: | 56 |
| NDAs: | 66 |
| Patent Litigation for Bionpharma: | See patent lawsuits for Bionpharma |
Drugs and US Patents for Bionpharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bionpharma | VALPROIC ACID | valproic acid | CAPSULE;ORAL | 073484-001 | Jun 29, 1993 | AB | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bionpharma | METHIMAZOLE | methimazole | TABLET;ORAL | 218149-002 | Sep 25, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bionpharma | STAVZOR | valproic acid | CAPSULE, DELAYED RELEASE;ORAL | 022152-003 | Jul 29, 2008 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bionpharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bionpharma | MIDOL LIQUID GELS | ibuprofen | CAPSULE;ORAL | 021472-001 | Oct 18, 2002 | 6,251,426 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Bionpharma Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 101166520 | ⤷ Try a Trial |
| Mexico | 2007011039 | ⤷ Try a Trial |
| China | 102940887 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bionpharma Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1613288 | C300497 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: FINGOLIMOD ALSMEDE FARMACEUTISCH AANVAARDBARE AFGELEIDEN DAARVAN, IN HET BIJZONDER FINGOLIMOD HYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/11/677/001-004 20110317 |
| 2782584 | 21C1058 | France | ⤷ Try a Trial | PRODUCT NAME: COMPOSITION CONTENANT A LA FOIS DE L'ESTRADIOL (17SS-ESTRADIOL), Y COMPRIS SOUS FORME HEMIHYDRATEE, ET DE LA PROGESTERONE; NAT. REGISTRATION NO/DATE: NL51886 20210421; FIRST REGISTRATION: BE - BE582231 20210406 |
| 0162036 | C300028 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LEVETIRACETAM; REGISTRATION NO/DATE: EU/1/00/146/001 - EU/1/00/146/026 20000929 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.