Axsome Malta Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AXSOME MALTA, and what generic alternatives to AXSOME MALTA drugs are available?
AXSOME MALTA has one approved drug.
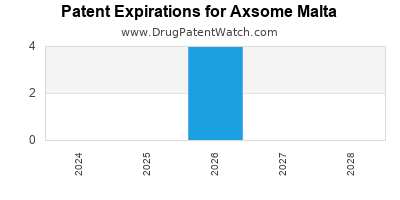
There are twenty-seven US patents protecting AXSOME MALTA drugs.
There are fifty-nine patent family members on AXSOME MALTA drugs in twenty-two countries and thirteen supplementary protection certificates in twelve countries.
Drugs and US Patents for Axsome Malta
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Axsome Malta | SUNOSI | solriamfetol hydrochloride | TABLET;ORAL | 211230-001 | Jun 17, 2019 | RX | Yes | No | 11,753,368 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Axsome Malta | SUNOSI | solriamfetol hydrochloride | TABLET;ORAL | 211230-002 | Jun 17, 2019 | RX | Yes | Yes | 10,512,609 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Axsome Malta | SUNOSI | solriamfetol hydrochloride | TABLET;ORAL | 211230-002 | Jun 17, 2019 | RX | Yes | Yes | 11,648,232 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Axsome Malta | SUNOSI | solriamfetol hydrochloride | TABLET;ORAL | 211230-002 | Jun 17, 2019 | RX | Yes | Yes | 11,160,779 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Axsome Malta | SUNOSI | solriamfetol hydrochloride | TABLET;ORAL | 211230-001 | Jun 17, 2019 | RX | Yes | No | 10,912,754 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Axsome Malta | SUNOSI | solriamfetol hydrochloride | TABLET;ORAL | 211230-001 | Jun 17, 2019 | RX | Yes | No | 11,850,227 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Axsome Malta Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Denmark | 3509581 | ⤷ Try a Trial |
| Netherlands | 301037 | ⤷ Try a Trial |
| European Patent Office | 1890684 | ⤷ Try a Trial |
| South Korea | 20230145525 | ⤷ Try a Trial |
| South Korea | 102513800 | ⤷ Try a Trial |
| Mexico | 2019014409 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Axsome Malta Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1890684 | 14/2020 | Austria | ⤷ Try a Trial | PRODUCT NAME: SOLRIAMFETOL ODER EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ DAVON, VORZUGSWEISE EIN HYDROCHLORIDSALZ DAVON; REGISTRATION NO/DATE: EU/1/19/1408 (MITTEILUNG) 20200120 |
| 1890684 | 2090011-4 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SOLRIAMFETOL OR A PHARMACEUTICALLY ACCEPTABLE SALT THEROF, PREFERABLY A HYDROCHLORIDE SALT THEROF; REG. NO/DATE: EU/1/19/1408 20200120 |
| 1890684 | CR 2020 00016 | Denmark | ⤷ Try a Trial | PRODUCT NAME: SOLRIAMFETOL ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, FORTRINSVIS ET HYDROCHLORIDSALT DERAF; REG. NO/DATE: EU/1/19/1408 20200120 |
| 1890684 | C01890684/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: SOLRIAMFETOLUM; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 68177 22.03.2022 |
| 1890684 | 132020000000040 | Italy | ⤷ Try a Trial | PRODUCT NAME: SOLRIAMFETOL O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE, PREFERIBILMENTE UN SUO SALE CLORIDRATO(SUNOSI); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/19/1408, 20200120 |
| 1890684 | CA 2020 00016 | Denmark | ⤷ Try a Trial | PRODUCT NAME: SOLRIAMFETOL ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, FORTRINSVIST ET HYDROCHLORIDSALT DERAF; REG. NO/DATE: EU/1/19/1408 20200120 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.