Almirall Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ALMIRALL, and what generic alternatives to ALMIRALL drugs are available?
ALMIRALL has twelve approved drugs.
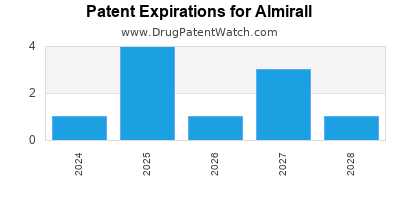
There are twenty US patents protecting ALMIRALL drugs.
There are two hundred and eleven patent family members on ALMIRALL drugs in thirty-one countries and thirty-one supplementary protection certificates in fourteen countries.
Drugs and US Patents for Almirall
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Almirall | CORDRAN | flurandrenolide | LOTION;TOPICAL | 013790-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | 7,300,931 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | 10,669,236 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Almirall | SOLAGE | mequinol; tretinoin | SOLUTION;TOPICAL | 020922-001 | Dec 10, 1999 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | 10,323,001 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Almirall | KLISYRI | tirbanibulin | OINTMENT;TOPICAL | 213189-001 | Dec 14, 2020 | RX | Yes | Yes | 8,236,799 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Almirall
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Almirall | SOLAGE | mequinol; tretinoin | SOLUTION;TOPICAL | 020922-001 | Dec 10, 1999 | 5,194,247 | ⤷ Try a Trial |
| Almirall | AZELEX | azelaic acid | CREAM;TOPICAL | 020428-001 | Sep 13, 1995 | 4,386,104 | ⤷ Try a Trial |
| Almirall | SOLAGE | mequinol; tretinoin | SOLUTION;TOPICAL | 020922-001 | Dec 10, 1999 | 5,470,567 | ⤷ Try a Trial |
| Almirall | ALTABAX | retapamulin | OINTMENT;TOPICAL | 022055-001 | Apr 12, 2007 | RE39128 | ⤷ Try a Trial |
| Almirall | VERDESO | desonide | AEROSOL, FOAM;TOPICAL | 021978-001 | Sep 19, 2006 | 6,730,288 | ⤷ Try a Trial |
| Almirall | ACZONE | dapsone | GEL;TOPICAL | 207154-001 | Feb 24, 2016 | 6,620,435 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ALMIRALL drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Gel | 7.5% | ➤ Subscribe | 2017-02-13 |
International Patents for Almirall Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2014167027 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2010135429 | ⤷ Try a Trial |
| European Patent Office | 2438910 | ⤷ Try a Trial |
| Cyprus | 1114825 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2008079339 | ⤷ Try a Trial |
| Japan | 2011093914 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Almirall Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1304992 | 2013C/060 | Belgium | ⤷ Try a Trial | PRODUCT NAME: CLINDAMYCINE (ALS CLINDAMYCINE FOSFAAT) EN TRETINOINE; AUTHORISATION NUMBER AND DATE: BE437507 20130506 |
| 1836169 | 2021C/001 | Belgium | ⤷ Try a Trial | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), ASSIGNMENT |
| 2435024 | 21C1020 | France | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE FORMOTEROL (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI), GLYCOPYRROLATE (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI) ET BUDESONIDE (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI); REGISTRATION NO/DATE: EU/1/20/1498 20201210 |
| 1836169 | C202130061 | Spain | ⤷ Try a Trial | PRODUCT NAME: TIRBANIBULINA; NATIONAL AUTHORISATION NUMBER: EU/1/21/1558; DATE OF AUTHORISATION: 20210716; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/21/1558; DATE OF FIRST AUTHORISATION IN EEA: 20210716 |
| 1836169 | 49/2021 | Austria | ⤷ Try a Trial | PRODUCT NAME: TIRBANIBULIN ODER EIN SALZ, SOLVAT ODER HYDRAT DAVON; REGISTRATION NO/DATE: EU/1/21/1558 (MITTEILUNG) 20210719 |
| 1836169 | CA 2021 00042 | Denmark | ⤷ Try a Trial | PRODUCT NAME: TIRBANIBULIN, ELLER ET SALT, SOLVAT ELLER HYDRAT DERAF; REG. NO/DATE: EU/1/21/1558 20210719 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.