Almatica Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ALMATICA, and what generic alternatives to ALMATICA drugs are available?
ALMATICA has nine approved drugs.
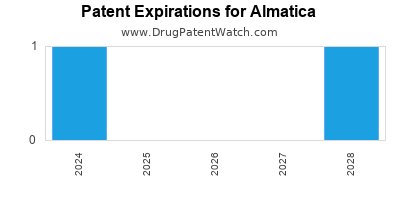
There are three US patents protecting ALMATICA drugs.
There are fifty-three patent family members on ALMATICA drugs in thirty-one countries and eighteen supplementary protection certificates in nine countries.
Drugs and US Patents for Almatica
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Almatica | ZESTORETIC | hydrochlorothiazide; lisinopril | TABLET;ORAL | 019888-001 | Sep 20, 1990 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Almatica | MACROBID | nitrofurantoin; nitrofurantoin, macrocrystalline | CAPSULE;ORAL | 020064-001 | Dec 24, 1991 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Almatica | GRALISE | gabapentin | TABLET;ORAL | 022544-002 | Jan 28, 2011 | AB2 | RX | Yes | No | 7,438,927 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Almatica | MACRODANTIN | nitrofurantoin, macrocrystalline | CAPSULE;ORAL | 016620-002 | Approved Prior to Jan 1, 1982 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Almatica | MACRODANTIN | nitrofurantoin, macrocrystalline | CAPSULE;ORAL | 016620-003 | Approved Prior to Jan 1, 1982 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Almatica | LOREEV XR | lorazepam | CAPSULE, EXTENDED RELEASE;ORAL | 214826-003 | Aug 27, 2021 | RX | Yes | Yes | 8,999,393 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Almatica
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Almatica | ZESTORETIC | hydrochlorothiazide; lisinopril | TABLET;ORAL | 019888-001 | Sep 20, 1990 | 4,472,380 | ⤷ Try a Trial |
| Almatica | GRALISE | gabapentin | TABLET;ORAL | 022544-001 | Jan 28, 2011 | 6,723,340 | ⤷ Try a Trial |
| Almatica | VENLAFAXINE BESYLATE | venlafaxine besylate | TABLET, EXTENDED RELEASE;ORAL | 215429-001 | Jun 29, 2022 | 6,717,015 | ⤷ Try a Trial |
| Almatica | ZESTORETIC | hydrochlorothiazide; lisinopril | TABLET;ORAL | 019888-002 | Jul 20, 1989 | 4,374,829*PED | ⤷ Try a Trial |
| Almatica | GRALISE | gabapentin | TABLET;ORAL | 022544-002 | Jan 28, 2011 | 8,333,992 | ⤷ Try a Trial |
| Almatica | MACROBID | nitrofurantoin; nitrofurantoin, macrocrystalline | CAPSULE;ORAL | 020064-001 | Dec 24, 1991 | 4,772,473 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ALMATICA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 300 mg and 600 mg | ➤ Subscribe | 2011-10-31 |
Premature patent expirations for ALMATICA
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for Almatica Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2007079195 | ⤷ Try a Trial |
| Philippines | 12015501534 | ⤷ Try a Trial |
| South Korea | 20150118119 | ⤷ Try a Trial |
| Japan | 2008508317 | ⤷ Try a Trial |
| Ukraine | 117233 | ⤷ Try a Trial |
| European Patent Office | 1976503 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Almatica Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2236132 | CA 2015 00004 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ZOLPIDEM OG FARMACEUTISK ACCEPTABLE SALTE HERAF, HERUNDER ZOLPIDEMTARTRAT; NAT. REG. NO/DATE: 47607 OG 47608 20120719; FIRST REG. NO/DATE: (B BE424286 OG BE424295 20120718 |
| 0347066 | C300155 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: ESCITALOPRAM, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVA ARDBAAR ZUURADDITIEZOUT, IN HET BIJZONDER ESCITALOPRAMOXALAAT; REGISTRATION NO/DATE: RVG 30490 - RVG 30497 20040427 |
| 0454511 | 99C0009 | Belgium | ⤷ Try a Trial | PRODUCT NAME: IRBESARTAN / HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/98/086/001 19981015 |
| 2236132 | 92636 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: ZOLPIDEM ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES |
| 0503785 | CA 2011 00026 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF OLMESARTAN MEDOXOMIL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND AMLODIPINE BESYLATE AND HYDROCHLOROTHIAZIDE; NAT. REG. NO/DATE: 46260-46269 (DK) 20110323; FIRST REG. NO/DATE: DE 79810.00.00 20101216 |
| 2236132 | 122015000006 | Germany | ⤷ Try a Trial | PRODUCT NAME: ZOLPIDEM UND PHARMAZEUTISCH VERTRAEGLICHE SALZE DAVON; NAT. REGISTRATION NO/DATE: 83439.00.00 83440.00.00 20120725 FIRST REGISTRATION: BELGIEN BE424286 BE424295 20120718 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |