Actelion Company Profile
✉ Email this page to a colleague
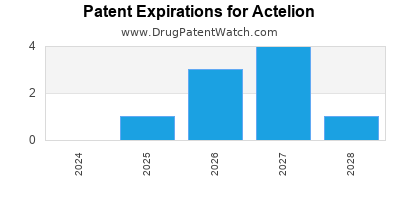
What is the competitive landscape for ACTELION, and when can generic versions of ACTELION drugs launch?
ACTELION has nine approved drugs.
There are fifteen US patents protecting ACTELION drugs.
There are two hundred and forty-eight patent family members on ACTELION drugs in thirty-nine countries and thirty-four supplementary protection certificates in seventeen countries.
Summary for Actelion
| International Patents: | 248 |
| US Patents: | 15 |
| Tradenames: | 7 |
| Ingredients: | 7 |
| NDAs: | 9 |
| PTAB Cases with Actelion as petitioner: | See PTAB cases with Actelion as petitioner |
Drugs and US Patents for Actelion
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Actelion | UPTRAVI | selexipag | POWDER;INTRAVENOUS | 214275-001 | Jul 29, 2021 | RX | Yes | Yes | 7,205,302 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Actelion | UPTRAVI | selexipag | TABLET;ORAL | 207947-002 | Dec 21, 2015 | AB | RX | Yes | Yes | 9,173,881 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Actelion | UPTRAVI | selexipag | TABLET;ORAL | 207947-004 | Dec 21, 2015 | AB | RX | Yes | No | 9,173,881 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Actelion
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Actelion | TRACLEER | bosentan | TABLET;ORAL | 021290-001 | Nov 20, 2001 | 5,292,740 | ⤷ Try a Trial |
| Actelion | ZAVESCA | miglustat | CAPSULE;ORAL | 021348-001 | Jul 31, 2003 | 5,472,969 | ⤷ Try a Trial |
| Actelion | TRACLEER | bosentan | TABLET;ORAL | 021290-002 | Nov 20, 2001 | 5,292,740 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ACTELION drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | for Oral Suspension | 32 mg | ➤ Subscribe | 2019-02-08 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2017-10-18 |
International Patents for Actelion Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 108289890 | ⤷ Try a Trial |
| Spain | 2652590 | ⤷ Try a Trial |
| Hungary | E026166 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Actelion Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0526708 | C300097 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BOSENTAN, DESGEWENST IN DE VORM VAN EEN ZOUT OF EEN HYDRAAT OF IN DE VORM VAN EEN ESTER VAN DE HYDROXYLGROEP VAN DE 2-HYDROXYETHOXY REST MET EEN ZUUR MET DE FORMULE R5-OH, WAARIN R5 EEN C1-7-ALKANOYL, BENZOYL, OF HETEROCYCLYCARBONYL VOORSTELT; NATL. REGISTRATION NO/DATE: U/1/02/220/001 - 005 20020515; FIRST REGISTRATION: CH IKS 58841 01 - 02 20020228 |
| 1345920 | SPC/GB14/018 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MACITENTAN, WHICH IS N-(5-(4-BROMOPHENYL)-6-(2((5-BROMOPYRIMIDIN-2-YL)OXY)ETHOXY)PYRIMIDIN-4-YL)-N'-PROPYLSULFURIC DIAMIDE; REGISTERED: UK EU/1/13/893 20131220 |
| 1400518 | 93266 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: SELEXIPAG OU UN SEL DE CELUI-CI; FIRST REGISTRATION DATE: 20160519 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.