TEVA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TEVA, and when can generic versions of TEVA drugs launch?
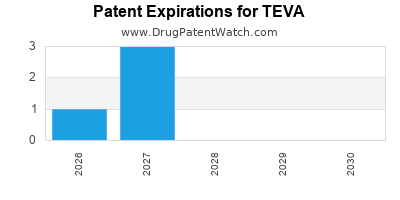
TEVA has seven hundred and thirty-six approved drugs.
There are one hundred US patents protecting TEVA drugs. There are fifty-three tentative approvals on TEVA drugs.
There are nine hundred and thirteen patent family members on TEVA drugs in forty-nine countries and one thousand and fifty-six supplementary protection certificates in eighteen countries.
Summary for TEVA
| International Patents: | 913 |
| US Patents: | 100 |
| Tradenames: | 508 |
| Ingredients: | 443 |
| NDAs: | 736 |
| Patent Litigation for TEVA: | See patent lawsuits for TEVA |
| PTAB Cases with TEVA as petitioner: | See PTAB cases with TEVA as petitioner |
Drugs and US Patents for TEVA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Parenteral | ERYTHROMYCIN LACTOBIONATE | erythromycin lactobionate | INJECTABLE;INJECTION | 063253-002 | Jul 30, 1993 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Teva Branded Pharm | AUSTEDO | deutetrabenazine | TABLET;ORAL | 208082-003 | Apr 3, 2017 | RX | Yes | Yes | 11,813,232*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Teva Pharms Usa Inc | ELIGLUSTAT TARTRATE | eliglustat tartrate | CAPSULE;ORAL | 212474-001 | Dec 27, 2021 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Teva | INDAPAMIDE | indapamide | TABLET;ORAL | 074665-002 | Apr 4, 1997 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Teva Pharms | METHYLPHENIDATE HYDROCHLORIDE | methylphenidate hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 078873-002 | Jul 19, 2012 | AB2 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva | AUSTEDO XR | deutetrabenazine | TABLET, EXTENDED RELEASE;ORAL | 216354-002 | Feb 17, 2023 | RX | Yes | No | 11,179,386*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Teva | ALBUTEROL SULFATE | albuterol sulfate | TABLET;ORAL | 072780-001 | Jun 25, 1993 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TEVA
Paragraph IV (Patent) Challenges for TEVA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 5 mg, 10 mg, 20 mg, 30 mg | ➤ Subscribe | 2009-11-18 |
| ➤ Subscribe | Injection | 40 mg/mL, 1 mL pre-filled syringe | ➤ Subscribe | 2014-02-26 |
| ➤ Subscribe | Tablets | 0.15 mg/0.03 mg/0.01 mg | ➤ Subscribe | 2008-01-22 |
| ➤ Subscribe | Tablets | 0.1 mg/0.02 mg and 0.01 mg | ➤ Subscribe | 2009-11-16 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | for Injection | 3.5 mg/vial | ➤ Subscribe | 2016-10-26 |
| ➤ Subscribe | Tablets | 0.5 mg and 1 mg | ➤ Subscribe | 2010-05-17 |
| ➤ Subscribe | Extended-release Capsule | 15 mg and 30 mg | ➤ Subscribe | 2008-08-11 |
| ➤ Subscribe | Injection | 40 mg/mL, 1 mL pre-filled syringe | ➤ Subscribe | 2014-01-29 |
| ➤ Subscribe | Tablets | 0.15 mg/0.03 mg | ➤ Subscribe | 2004-03-29 |
| ➤ Subscribe | Tablets | 0.15 mg/0.02 mg, 0.15 mg/0.025 mg, 0.15 mg/0.03 mg and 0.01 mg | ➤ Subscribe | 2013-07-10 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Tablets | 1 mg/0.02 mg and 75 mg | ➤ Subscribe | 2006-04-17 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
Premature patent expirations for TEVA
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for TEVA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Singapore | 11201908477Q | ⤷ Try a Trial |
| Hong Kong | 1212232 | ⤷ Try a Trial |
| Japan | 2007517902 | ⤷ Try a Trial |
| Denmark | 2436414 | ⤷ Try a Trial |
| Japan | 2021000468 | ⤷ Try a Trial |
| Japan | 2015528516 | ⤷ Try a Trial |
| New Zealand | 627544 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for TEVA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2962690 | C20190029 00295 | Estonia | ⤷ Try a Trial | PRODUCT NAME: APREMILAST;REG NO/DATE: EU/1/14/981 16.01.2015 |
| 2635588 | C02635588/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: VELPATASVIR + SOFOSBUVIR + VOXILAPREVIR; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66510 08.12.2017 |
| 0503785 | 9/2011 | Austria | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION AUS OLMESARTAN MEDOXOMIL, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH AKZEPTABLEN SALZES, AMLODIPIN BESILAT UND HYDROCHLOROTHIAZID; NAT. REGISTRATION NO/DATE: 1-30068 - 1-30072 20110216; FIRST REGISTRATION: DE 79810.00.00 - 79814.00.00, UND 79815.00.00 - 79819.00.00 20101216 |
| 0810209 | C300283 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: DARUNAVIR, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF ESTER, IN HET BIJZONDER DARUNAVIR ETHANOLAAT; REGISTRATION NO/DATE: EU/1/06/380/001 20070212 |
| 0627406 | 1190015-6 | Sweden | ⤷ Try a Trial | PRODUCT NAME: FINGOLIMOD; REG. NO/DATE: EU/1/11/677/001-04 20110317 |
| 0273658 | C300171 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: DULOXETINE EN FARMACEUTISCH AANVAARDBARE ZUURADDITIEZOUTEN DAARVAN, EN IN HET BIJZONDER DULOXETINE HYDROCHLORIDE; NATL REGISTRATION NO/DATE: EU/1/04/280/001-006 20040811 |
| 0915894 | C00915894/02 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: TENOFOVIRDISOPROXIL + EFAVIRENZ + EMTRICITABIN; REGISTRATION NUMBER/DATE: SWISSMEDIC 60011 20.11.2009 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.