OTSUKA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for OTSUKA, and what generic alternatives to OTSUKA drugs are available?
OTSUKA has seventeen approved drugs.
There are fifty-six US patents protecting OTSUKA drugs.
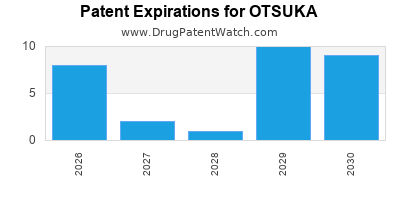
There are one thousand and seventeen patent family members on OTSUKA drugs in fifty-four countries and forty-nine supplementary protection certificates in sixteen countries.
Summary for OTSUKA
| International Patents: | 1017 |
| US Patents: | 56 |
| Tradenames: | 15 |
| Ingredients: | 10 |
| NDAs: | 17 |
| Patent Litigation for OTSUKA: | See patent lawsuits for OTSUKA |
Drugs and US Patents for OTSUKA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Otsuka | RAXAR | grepafloxacin hydrochloride | TABLET;ORAL | 020695-003 | May 14, 1998 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Otsuka | REXULTI | brexpiprazole | TABLET;ORAL | 205422-003 | Jul 10, 2015 | AB | RX | Yes | No | RE48059 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Otsuka | ABILIFY | aripiprazole | TABLET;ORAL | 021436-004 | Nov 15, 2002 | AB | RX | Yes | No | 9,125,939 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-006 | Nov 13, 2017 | RX | Yes | No | 8,956,288 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | JYNARQUE | tolvaptan | TABLET;ORAL | 204441-004 | Apr 23, 2018 | RX | Yes | No | 10,905,694 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-006 | Nov 13, 2017 | RX | Yes | No | 8,258,962 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Otsuka | ABILIFY | aripiprazole | TABLET, ORALLY DISINTEGRATING;ORAL | 021729-002 | Jun 7, 2006 | DISCN | Yes | No | 8,759,350 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OTSUKA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Otsuka Pharm Co Ltd | ABILIFY MAINTENA KIT | aripiprazole | FOR SUSPENSION, EXTENDED RELEASE;INTRAMUSCULAR | 202971-004 | Sep 29, 2014 | 5,006,528*PED | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET, ORALLY DISINTEGRATING;ORAL | 021729-004 | Jun 7, 2006 | 8,642,760*PED | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET, ORALLY DISINTEGRATING;ORAL | 021729-002 | Jun 7, 2006 | 9,089,567 | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET;ORAL | 021436-003 | Nov 15, 2002 | 9,387,182 | ⤷ Try a Trial |
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-002 | Nov 13, 2017 | 8,642,760 | ⤷ Try a Trial |
| Otsuka | ABILIFY | aripiprazole | TABLET;ORAL | 021436-003 | Nov 15, 2002 | 8,580,796*PED | ⤷ Try a Trial |
| Otsuka | ABILIFY MYCITE KIT | aripiprazole | TABLET;ORAL | 207202-003 | Nov 13, 2017 | 9,089,567 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for OTSUKA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 60 mg | ➤ Subscribe | 2018-03-26 |
| ➤ Subscribe | Tablets | 2 mg, 5 mg, 10 mg, 15 mg, 20 mg and 30 mg | ➤ Subscribe | 2006-11-15 |
| ➤ Subscribe | Tablets | 15 mg and 30 mg | ➤ Subscribe | 2013-09-23 |
| ➤ Subscribe | Injection | 6 mg/mL | ➤ Subscribe | 2012-12-26 |
| ➤ Subscribe | Oral Solution | 1 mg/mL | ➤ Subscribe | 2007-12-20 |
| ➤ Subscribe | Orally Disintegrating Tablets | 10 mg, 15 mg, 20 mg and 30 mg | ➤ Subscribe | 2006-11-15 |
International Patents for OTSUKA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | PA03000440 | ⤷ Try a Trial |
| Russian Federation | 2005140285 | ⤷ Try a Trial |
| China | 100335047 | ⤷ Try a Trial |
| Malaysia | 142746 | ⤷ Try a Trial |
| China | 104376659 | ⤷ Try a Trial |
| Japan | 5614991 | ⤷ Try a Trial |
| Russian Federation | 2012143791 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for OTSUKA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2207786 | 2023C/550 | Belgium | ⤷ Try a Trial | PRODUCT NAME: CEDAZURIDINE, OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI; AUTHORISATION NUMBER AND DATE: EU/1/23/1756 20230918 |
| 1869025 | LUC00086 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: BREXPIPRAZOLE OU UN SEL DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/18/1294 20180730 |
| 1869025 | 300946 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BREXPIPRAZOLE, DESGEWENST IN DE VORM VAN EEN ZOUT; REGISTRATION NO/DATE: EU/1/18/1294 20180727 |
| 1675573 | 300669 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: ARIPIPRAZOLE; REGISTRATION NO/DATE: EU/1/13/882 20131115 |
| 2207786 | 301257 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: SAMENSTELLING WELKE OMVAT: CEDAZURIDINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; EN DECITABINE; REGISTRATION NO/DATE: EU/1/23/1756 20230918 |
| 1675573 | PA2014020 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: ARIPIPRAZOLUM; REGISTRATION NO/DATE: EU/1/13/882 20131115 |
| 1869025 | C01869025/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: BREXPIPRAZOL; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66475 17.07.2018 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.