sodium nitrite Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Sodium Nitrite, and when can generic versions of Sodium Nitrite launch?
Sodium Nitrite is a drug marketed by Hope Pharms and is included in one NDA. There are two patents protecting this drug.
This drug has twenty-seven patent family members in fourteen countries.
The generic ingredient in SODIUM NITRITE is sodium nitrite. There are one thousand four hundred and seventy-two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the sodium nitrite profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Sodium Nitrite
A generic version of sodium nitrite was approved as sodium nitrite by HOPE PHARMS on February 14th, 2012.
Summary for sodium nitrite
| International Patents: | 27 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 123 |
| Clinical Trials: | 58 |
| Patent Applications: | 1,397 |
| Formulation / Manufacturing: | see details |
| What excipients (inactive ingredients) are in sodium nitrite? | sodium nitrite excipients list |
| DailyMed Link: | sodium nitrite at DailyMed |
Recent Clinical Trials for sodium nitrite
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Alabama at Birmingham | Phase 1 |
| University of Alabama at Birmingham | Early Phase 1 |
| National Institutes of Health (NIH) | Phase 1 |
Medical Subject Heading (MeSH) Categories for sodium nitrite
Anatomical Therapeutic Chemical (ATC) Classes for sodium nitrite
US Patents and Regulatory Information for sodium nitrite
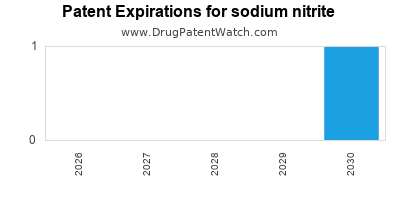
sodium nitrite is protected by three US patents.
Patents protecting sodium nitrite
Sodium nitrite-containing pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Sodium nitrite-containing pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: SODIUM NITRITE INJECTION IS ADMINISTERED BY INTRAVENOUS INJECTION
Sodium nitrite-containing pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: SODIUM NITRITE INJECTION IS INDICATED FOR SEQUENTIAL USE WITH SODIUM THIOSULFATE FOR THE TREATMENT OF ACUTE CYANIDE POISONING
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hope Pharms | SODIUM NITRITE | sodium nitrite | SOLUTION;INTRAVENOUS | 203922-001 | Feb 14, 2012 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Hope Pharms | SODIUM NITRITE | sodium nitrite | SOLUTION;INTRAVENOUS | 203922-001 | Feb 14, 2012 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for sodium nitrite
See the table below for patents covering sodium nitrite around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Portugal | 3569237 | ⤷ Try a Trial | |
| Japan | 5789519 | ⤷ Try a Trial | |
| Slovenia | 2395834 | ⤷ Try a Trial | |
| Japan | 2015199764 | 亜硝酸ナトリウムを含む医薬組成物 (SODIUM NITRITE-CONTAINING PHARMACEUTICAL COMPOSITIONS) | ⤷ Try a Trial |
| Japan | 6059297 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for sodium nitrite
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1912999 | 1490062-5 | Sweden | ⤷ Try a Trial | PRODUCT NAME: SIMEPREVIR, OR A SALT THEREOF, INCLUDING SIMEPREVIR SODIUM; REG. NO/DATE: EU/1/14/924 20140516 |
| 2932970 | SPC/GB18/041 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION COMPRISING DOLUTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF (E.G. DOLUTEGRAVIR SODIUM) AND RILPIVIRINE OR A PHARAMACEUTICALLY ACCEPTABLE SALT THEREOF (E.G. RILPIVIRINE HYDROCHLORIDE); REGISTERED: UK EU/1/18/1282 20180518; UK PLGB 35728/0055 20180518; UK PLGB 35728/0056 20180518; UK PLGB 35728/0057 20180518 |
| 2822954 | LUC00083 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR BICTEGRAVIR SODIUM; AUTHORISATION NUMBER AND DATE: EU/1/18/1289 20180625 |
| 1856135 | SPC/GB20/016 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: FOSTAMATINIB OR A PHARMACEUTICALLY ACCEPTABLE SALT OF FOSTAMATINIB, OR A HYDRATE, SOLVATE OR N-OXIDE OF FOSTAMATINIB OR THE PHARMACEUTICALLY ACCEPTABLE SALT OF FOSTAMATINIB, ESPECIALLY FOSTAMATINIB DISODIUM, OPTIONALLY IN FORM OF A HYDRATE; REGISTERED: UK EU/1/19/1405 (NI) 20200113; UK PLGB 12930/0022 20200113; UK PLGB 12930/0023 20200113 |
| 2203431 | 2015/009 | Ireland | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REGISTRATION NO/DATE: EU/1/14/983 20150115 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


