ingrezza Drug Patent Profile
✉ Email this page to a colleague
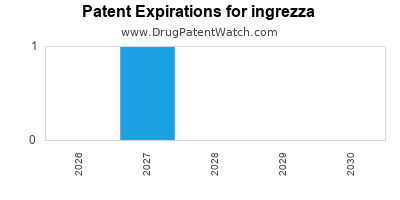
When do Ingrezza patents expire, and what generic alternatives are available?
Ingrezza is a drug marketed by Neurocrine and is included in two NDAs. There are twenty-two patents protecting this drug and two Paragraph IV challenges.
This drug has two hundred and seventy-six patent family members in thirty-six countries.
The generic ingredient in INGREZZA is valbenazine tosylate. One supplier is listed for this compound. Additional details are available on the valbenazine tosylate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Ingrezza
A generic version of ingrezza was approved as valbenazine tosylate by LUPIN LTD on April 5th, 2024.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ingrezza?
- What are the global sales for ingrezza?
- What is Average Wholesale Price for ingrezza?
Summary for ingrezza
| International Patents: | 276 |
| US Patents: | 22 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 50 |
| Clinical Trials: | 7 |
| Patent Applications: | 297 |
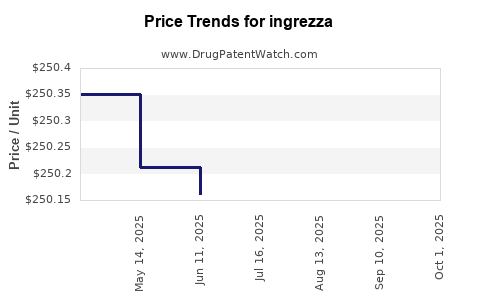
| Drug Prices: | Drug price information for ingrezza |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ingrezza |
| What excipients (inactive ingredients) are in ingrezza? | ingrezza excipients list |
| DailyMed Link: | ingrezza at DailyMed |
Recent Clinical Trials for ingrezza
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Neurocrine Biosciences | Phase 4 |
| Stephen Ruedrich | Phase 4 |
| Michael Bloch | Phase 2 |
Pharmacology for ingrezza
| Drug Class | Vesicular Monoamine Transporter 2 Inhibitor |
| Mechanism of Action | Vesicular Monoamine Transporter 2 Inhibitors |
Paragraph IV (Patent) Challenges for INGREZZA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| INGREZZA | Capsules | valbenazine tosylate | 60 mg | 209241 | 1 | 2022-02-14 |
| INGREZZA | Capsules | valbenazine tosylate | 40 mg and 80 mg | 209241 | 4 | 2021-04-12 |
US Patents and Regulatory Information for ingrezza
ingrezza is protected by thirty-three US patents and two FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neurocrine | INGREZZA | valbenazine tosylate | CAPSULE;ORAL | 209241-002 | Oct 4, 2017 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Neurocrine | INGREZZA | valbenazine tosylate | CAPSULE;ORAL | 209241-002 | Oct 4, 2017 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| Neurocrine | INGREZZA | valbenazine tosylate | CAPSULE;ORAL | 209241-003 | Apr 23, 2021 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Neurocrine | INGREZZA | valbenazine tosylate | CAPSULE;ORAL | 209241-001 | Apr 11, 2017 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| Neurocrine | INGREZZA SPRINKLE | valbenazine tosylate | CAPSULE;ORAL | 218390-002 | Apr 30, 2024 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Neurocrine | INGREZZA | valbenazine tosylate | CAPSULE;ORAL | 209241-002 | Oct 4, 2017 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for ingrezza
When does loss-of-exclusivity occur for ingrezza?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2819
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 18335259
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2020005373
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 76000
Estimated Expiration: ⤷ Start Trial
China
Patent: 1372567
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0250561
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 2090809
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 71873
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 3300
Patent: פורמולציית ולבנאזין במינון גבוה ותכשירים, שיטות וערכות קשורות להן (High dosage valbenazine formulation and compositions, methods, and kits related thereto)
Estimated Expiration: ⤷ Start Trial
Patent: 1770
Patent: פורמולציית ולבנאזין במינון גבוה ותכשירים, שיטות וערכות קשורות להן (High dosage valbenazine formulation and compositions, methods, and kits related thereto)
Estimated Expiration: ⤷ Start Trial
Patent: 9802
Patent: פורמולציית ולבנאזין במינון גבוה ותכשירים, שיטות וערכות קשורות להן (High dosage valbenazine formulation and compositions, methods, and kits related thereto)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 50006
Estimated Expiration: ⤷ Start Trial
Patent: 20534305
Patent: 高投与量バルベナジン製剤ならびにそれに関連する組成物、方法およびキット
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 20002841
Patent: FORMULACION DE VALBENAZINA DE ALTA DOSIS Y COMPOSICIONES, METODOS Y KITS RELACIONADOS CON LA MISMA. (HIGH DOSAGE VALBENAZINE FORMULATION AND COMPOSITIONS, METHODS, AND KITS RELATED THERETO.)
Estimated Expiration: ⤷ Start Trial
Moldova, Republic of
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 175
Patent: FORMULATION DE VALBENAZINE À DOSAGE ÉLEVÉ ET COMPOSITIONS, PROCÉDÉS ET KITS ASSOCIÉS
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 84333
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 02500305
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 714
Patent: FORMULACIJA VISOKE DOZE VALBENAZINA I SASTAVI, POSTUPCI I KOMPLETI POVEZANI SA NJIM (HIGH DOSAGE VALBENAZINE FORMULATION AND COMPOSITIONS, METHODS, AND KITS RELATED THERETO)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 84333
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 24958
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1919622
Patent: High dosage VALBENAZINE formulation and compositions, methods, and kits related thereto
Estimated Expiration: ⤷ Start Trial
Patent: 75681
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ingrezza around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| South Korea | 20250020722 | ⤷ Start Trial | |
| South Korea | 20200066662 | ⤷ Start Trial | |
| Canada | 3051829 | ⤷ Start Trial | |
| Taiwan | 201919622 | High dosage VALBENAZINE formulation and compositions, methods, and kits related thereto | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2018140095 | ⤷ Start Trial | |
| Japan | 2020514318 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
INGREZZA: Market Landscape and Financial Outlook
More… ↓
DrugChatter Q&A for INGREZZA
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.