Last updated: February 19, 2026
Zolpidem tartrate, a nonbenzodiazepine hypnotic, maintains a significant presence in the insomnia treatment market. Its market performance is driven by patent expirations, generic competition, and evolving regulatory landscapes. This analysis details the drug's intellectual property status, market size, competitive environment, and projected financial trajectory, offering insights for R&D and investment strategies.
What is the Current Intellectual Property Landscape for Zolpidem Tartrate?
The foundational patents for zolpidem tartrate have long expired, significantly opening the market to generic manufacturers. The original patent for zolpidem tartrate was granted to Sanofi-Aventis (now Sanofi) in the late 1980s and early 1990s. Key patents, such as U.S. Patent No. 4,794,190 and others related to its manufacturing processes and formulations, expired in the mid-2000s.
- Original Patent Expiration: The primary composition of matter patents for zolpidem expired around 2005-2006.
- Formulation Patents: While some later formulation patents may have had staggered expirations, the core intellectual property protection is no longer in place for standard immediate-release and extended-release formulations.
- Authorized Generics: The absence of strong patent protection allows for the widespread availability of authorized generics and multi-source generics, leading to intense price competition.
What is the Current Market Size and Growth Potential for Zolpidem Tartrate?
The global market for zolpidem tartrate, while mature, continues to represent a substantial revenue stream due to its widespread use for the short-term treatment of insomnia.
- Market Value: The global sleep aid market, of which zolpidem tartrate is a significant component, was valued at approximately USD 63.4 billion in 2022 [1]. While specific figures for zolpidem tartrate alone are not always isolated in market reports, it is understood to be a multi-billion dollar segment within this broader market.
- Growth Drivers:
- Aging Population: The increasing prevalence of sleep disorders in older adults is a consistent driver of demand.
- Increased Awareness: Greater public and medical awareness of the impact of chronic insomnia on overall health contributes to prescription volume.
- Lifestyle Factors: Stress, shift work, and other modern lifestyle factors exacerbate insomnia, sustaining demand.
- Growth Restraints:
- Generic Competition: Price erosion due to a large number of generic competitors limits overall market value growth.
- Side Effect Profile: Concerns regarding next-day drowsiness, dependence, and potential for abuse (though lower than benzodiazepines) lead some prescribers and patients to seek alternatives.
- Regulatory Scrutiny: Monitoring and potential limitations on prescription due to the risk of misuse or dependence.
Projected Market Growth (Sleep Aids - General Segment): The broader sleep aid market is projected to grow at a compound annual growth rate (CAGR) of 6.5% from 2023 to 2030, reaching an estimated value of over USD 100 billion [1]. Zolpidem tartrate's share within this growth will likely be constrained by generic pricing pressures but supported by its established efficacy and physician familiarity.
Who are the Key Players in the Zolpidem Tartrate Market?
The market for zolpidem tartrate is characterized by a fragmented landscape dominated by generic pharmaceutical manufacturers.
- Original Innovator: Sanofi was the original developer and marketer of zolpidem tartrate under the brand name Ambien.
- Major Generic Manufacturers: Numerous companies produce and distribute generic versions of zolpidem tartrate. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cipla Ltd.
- Mylan N.V. (now part of Viatris)
- Apotex Inc.
- Authorized Generics: Branded pharmaceutical companies may also market their own generic versions (authorized generics) to compete directly with third-party generics, often at lower price points than their branded counterparts.
- Contract Manufacturers: Many smaller or regional players rely on contract manufacturing organizations (CMOs) for production.
The competitive dynamic is primarily driven by cost-efficiency and supply chain reliability rather than product differentiation, as the active pharmaceutical ingredient (API) and core formulations are widely available.
What are the Regulatory Considerations for Zolpidem Tartrate?
Zolpidem tartrate is subject to stringent regulatory oversight due to its potential for misuse and dependence.
- Scheduling: In the United States, zolpidem is classified as a Schedule IV controlled substance by the Drug Enforcement Administration (DEA) [2]. This classification imposes specific requirements for prescription, dispensing, and record-keeping.
- FDA Labeling: The U.S. Food and Drug Administration (FDA) has issued several safety communications and label updates for zolpidem products, emphasizing the risks of serious injury and death from sleepwalking and other complex sleep-related behaviors, as well as impairment from residual morning effects. These updates have led to recommendations for lower starting doses, particularly for women and older adults [3].
- International Regulations: Similar scheduling and safety monitoring are in place in many other countries, impacting prescribing practices and market access.
- Abuse Deterrence: While not a primary focus for zolpidem as it is for opioids, regulatory bodies are increasingly scrutinizing all controlled substances for potential abuse-deterrent formulations or risk mitigation strategies, although no major developments specific to zolpidem have emerged in this area beyond dosage adjustments and warnings.
What is the Financial Trajectory and Profitability of Zolpidem Tartrate Products?
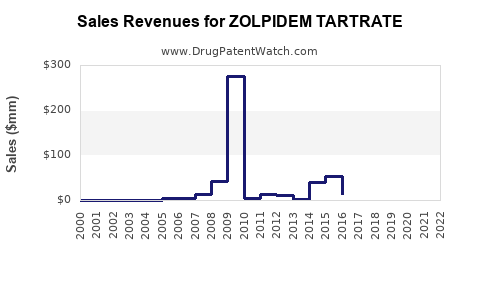
The financial trajectory of zolpidem tartrate is characterized by high initial revenue during patent exclusivity, followed by a steep decline and stabilization in the generic market.
- Branded Revenue: Peak annual sales for Ambien reached over USD 2 billion in the U.S. before significant generic entry [4].
- Generic Market Dynamics:
- Price Erosion: The introduction of generics leads to rapid price declines, often exceeding 80-90% from the branded price.
- Volume-Driven Profitability: Profitability for generic manufacturers relies on achieving high production volumes at low manufacturing costs and securing favorable market share through bidding on tenders or distribution agreements.
- Cost of Goods Sold (COGS): Efficient sourcing of zolpidem tartrate API and streamlined manufacturing processes are critical for maintaining margins.
- Gross Margins: Gross margins on generic drugs are typically lower than for patented products, often in the range of 20-40%, depending on market competition and scale.
- Net Margins: Net profit margins for generic manufacturers can vary significantly but are generally lower than branded pharmaceuticals, often ranging from 5-15%, after accounting for R&D (for lifecycle management), marketing, sales, and administrative expenses.
- Market Stabilization: The market tends to stabilize once the number of generic competitors reaches a certain equilibrium, and pricing finds a new, lower baseline. Demand remains relatively stable due to the drug's established clinical utility.
- Opportunities for Generic Manufacturers:
- Cost Leadership: Focusing on ultra-low-cost manufacturing.
- Supply Chain Excellence: Ensuring consistent availability and reliable delivery.
- Specialized Formulations: Exploring niche formulations or combinations, though patentability is a significant hurdle post-expiration.
- Geographic Expansion: Targeting markets with less intense generic competition or different regulatory pathways.
What are the Future Prospects for Zolpidem Tartrate in the Pharmaceutical Landscape?
The future of zolpidem tartrate is largely defined by its status as a well-established, off-patent hypnotic agent.
- Continued Generic Dominance: The market will remain dominated by generic competition. Innovation will be primarily incremental, focusing on manufacturing efficiency and supply chain optimization rather than novel therapeutic advancements.
- Competition from Newer Hypnotics: While zolpidem remains a first-line option for many, newer classes of hypnotics and non-pharmacological treatments for insomnia will continue to compete for market share. These include orexin receptor antagonists (e.g., suvorexant, lemborexant), which offer different mechanisms of action and may have distinct side effect profiles.
- Evolving Sleep Disorder Management: A growing emphasis on cognitive behavioral therapy for insomnia (CBT-I) as a first-line treatment may temper the growth of pharmacological interventions over the long term.
- Lifecycle Management for Generics: Generic companies may focus on developing extended-release formulations or combination products if opportunities arise, but significant R&D investment is unlikely given the mature nature of the market.
- Regulatory Landscape: Ongoing pharmacovigilance and potential shifts in prescribing guidelines by regulatory bodies will continue to influence its usage. Any new safety signals could lead to further restrictions or warnings.
Key Takeaways
Zolpidem tartrate's market is characterized by mature intellectual property, intense generic competition, and stable but price-sensitive demand. Innovation has shifted from novel drug discovery to manufacturing efficiency and supply chain management for generic manufacturers. Regulatory oversight remains a critical factor, influencing prescribing practices and safety warnings. While newer sleep aids and non-pharmacological interventions are emerging, zolpidem tartrate's established efficacy and affordability ensure its continued, albeit highly competitive, market presence.
Frequently Asked Questions
-
What is the primary therapeutic indication for zolpidem tartrate?
Zolpidem tartrate is indicated for the short-term treatment of insomnia characterized by difficulties with sleep onset.
-
How has generic competition impacted the pricing of zolpidem tartrate?
Generic competition has led to significant price erosion, with generic versions trading at a fraction of the original branded drug's price.
-
What are the main safety concerns associated with zolpidem tartrate?
Key safety concerns include next-day impairment (drowsiness, cognitive impairment), complex sleep-related behaviors (e.g., sleepwalking), and potential for dependence or misuse.
-
Are there any new patent applications being filed for zolpidem tartrate itself?
Due to the age of the foundational patents, new patent applications for the basic zolpidem tartrate molecule are highly unlikely. Innovation would likely focus on novel delivery systems or combinations, which face high hurdles for patentability and market adoption in this mature space.
-
What is the future outlook for zolpidem tartrate in the context of evolving insomnia treatments?
Zolpidem tartrate will likely remain a significant generic option for insomnia due to its established efficacy and cost-effectiveness. However, it faces competition from newer pharmacological agents and a growing emphasis on non-pharmacological therapies like CBT-I.
Citations
[1] Grand View Research. (2023). Sleep Aids Market Size, Share & Trends Analysis Report By Product (Prescription Sleep Aids, OTC Sleep Aids), By Sleep Disorder (Insomnia, Sleep Apnea, Others), By Region, And Segment Forecasts, 2023 – 2030. https://www.grandviewresearch.com/industry-analysis/sleep-aids-market
[2] U.S. Drug Enforcement Administration. (n.d.). Schedules of Controlled Substances. https://www.dea.gov/drug-scheduling
[3] U.S. Food & Drug Administration. (2019, January 17). FDA Drug Safety Communication – FDA requires labeling changes for hypnotic drugs for insomnia to remove the higher recommended starting dose for women and to inform about other considerations. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-requires-labeling-changes-hypnotic-drugs-insomnia-remove-higher
[4] FiercePharma. (2007, August 21). Sanofi-Aventis losing insomnia sales as generics flood market. https://www.fiercepharma.com/marketing/sanofi-aventis-losing-insomnia-sales-generics-flood-market