ZIOPTAN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Zioptan, and what generic alternatives are available?
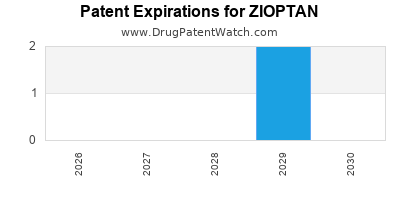
Zioptan is a drug marketed by Thea Pharma and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has eighty-five patent family members in twenty-six countries.
The generic ingredient in ZIOPTAN is tafluprost. There are three drug master file entries for this compound. Five suppliers are listed for this compound. Additional details are available on the tafluprost profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Zioptan
A generic version of ZIOPTAN was approved as tafluprost by MICRO LABS on August 19th, 2019.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ZIOPTAN?
- What are the global sales for ZIOPTAN?
- What is Average Wholesale Price for ZIOPTAN?
Summary for ZIOPTAN
| International Patents: | 85 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Patent Applications: | 882 |
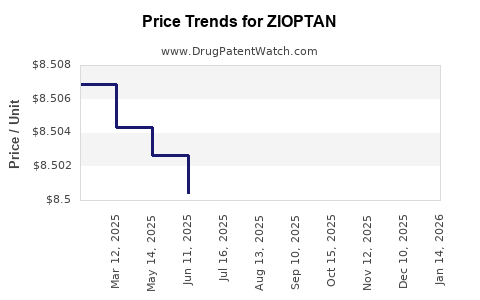
| Drug Prices: | Drug price information for ZIOPTAN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ZIOPTAN |
| What excipients (inactive ingredients) are in ZIOPTAN? | ZIOPTAN excipients list |
| DailyMed Link: | ZIOPTAN at DailyMed |
Pharmacology for ZIOPTAN
| Drug Class | Prostaglandin Analog |
| Mechanism of Action | Prostaglandin Receptor Agonists |
| Physiological Effect | Increased Prostaglandin Activity |
Paragraph IV (Patent) Challenges for ZIOPTAN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ZIOPTAN | Ophthalmic Solution | tafluprost | 0.0015% | 202514 | 2 | 2016-02-10 |
US Patents and Regulatory Information for ZIOPTAN
ZIOPTAN is protected by two US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Thea Pharma | ZIOPTAN | tafluprost | SOLUTION/DROPS;OPHTHALMIC | 202514-001 | Feb 10, 2012 | AT | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Thea Pharma | ZIOPTAN | tafluprost | SOLUTION/DROPS;OPHTHALMIC | 202514-001 | Feb 10, 2012 | AT | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ZIOPTAN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Thea Pharma | ZIOPTAN | tafluprost | SOLUTION/DROPS;OPHTHALMIC | 202514-001 | Feb 10, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ZIOPTAN
When does loss-of-exclusivity occur for ZIOPTAN?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1937
Patent: METODO Y COMPOSICION PARA TRATAR HIPERTENSION OCULAR Y GLAUCOMA QUE COMPRENDE ANALOGOS DE PGF-2ALFA, USO Y METODO PARA AUMENTAR LA SOLUBILIDAD ACUOSA Y MEJORAR LA ESTABILIDAD DE LOS ANALOGOS DE PGF-2ALFA
Estimated Expiration: ⤷ Start Trial
Patent: 0961
Patent: MÉTODO Y COMPOSICIÓN PARA TRATAR HIPERTENSIÓN OCULAR Y GLAUCOMA
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09252210
Patent: Method and composition for treating ocular hypertension and glaucoma
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0913109
Patent: solução aquosa oftálmica, uso de análogos de pgf2a, e, método para aumentar a solubilidade em água e melhorar a estabilidade de análogos de pgf2a em uma solução aquosa oftálmica
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 24194
Patent: PROCEDE ET COMPOSITION UTILISABLES POUR LE TRAITEMENT DE L'HYPERTENSION OCULAIRE ET DU GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 65185
Patent: PROCEDE ET COMPOSITION UTILISABLES POUR LE TRAITEMENT DE L'HYPERTENSION OCULAIRE ET DU GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
China
Patent: 2083413
Patent: Method and composition for treating ocular hypertension and glaucoma
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0140979
Estimated Expiration: ⤷ Start Trial
Patent: 0170769
Estimated Expiration: ⤷ Start Trial
Patent: 0200998
Estimated Expiration: ⤷ Start Trial
Patent: 0220361
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 15565
Estimated Expiration: ⤷ Start Trial
Patent: 20351
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 06977
Estimated Expiration: ⤷ Start Trial
Patent: 72249
Estimated Expiration: ⤷ Start Trial
Patent: 05334
Estimated Expiration: ⤷ Start Trial
Patent: 14877
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 3661
Patent: УПАКОВАННЫЙ РАСТВОР ДЛЯ ЛЕЧЕНИЯ ПОВЫШЕНИЯ ВНУТРИГЛАЗНОГО ДАВЛЕНИЯ И ГЛАУКОМЫ, ВКЛЮЧАЮЩИЙ АНАЛОГ PGF2α (PACKAGED SOLUTION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA, COMPRISING PGF2α ANALOGUE)
Estimated Expiration: ⤷ Start Trial
Patent: 1071413
Patent: СПОСОБ И КОМПОЗИЦИЯ ДЛЯ ЛЕЧЕНИЯ ПОВЫШЕНИЯ ВНУТРИГЛАЗНОГО ДАВЛЕНИЯ И ГЛАУКОМЫ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 27638
Patent: Procédé et composition pour traiter l'hypertension oculaire et le glaucome (Method and composition for treating ocular hypertension and glaucoma)
Estimated Expiration: ⤷ Start Trial
Patent: 06977
Patent: PROCÉDÉ ET COMPOSITION POUR TRAITER L'HYPERTENSION OCULAIRE ET LE GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 72249
Patent: Procédé et composition pour traiter l'hypertension oculaire et le glaucome (Method and composition for treating ocular hypertension and glaucoma)
Estimated Expiration: ⤷ Start Trial
Patent: 05334
Patent: PROCÉDÉ ET COMPOSITION POUR LE TRAITEMENT DE L'HYPERTENSION OCULAIRE ET DU GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 14877
Patent: PROCÉDÉ ET COMPOSITION POUR LE TRAITEMENT DE L'HYPERTENSION OCULAIRE ET DU GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 35656
Patent: PROCÉDÉ ET COMPOSITION POUR LE TRAITEMENT DE L'HYPERTENSION OCULAIRE ET DU GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 89446
Patent: PROCÉDÉ ET COMPOSITION POUR LE TRAITEMENT DE L'HYPERTENSION OCULAIRE ET DU GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 0156220
Patent: COMPOSITION TREATING OCULAR HYPERTENSION AND GLAUCOMA, AND USAGE THEREOF
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 33103
Estimated Expiration: ⤷ Start Trial
Patent: 49923
Estimated Expiration: ⤷ Start Trial
Patent: 58079
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 48317
Estimated Expiration: ⤷ Start Trial
Patent: 56868
Estimated Expiration: ⤷ Start Trial
Patent: 49992
Estimated Expiration: ⤷ Start Trial
Patent: 89789
Estimated Expiration: ⤷ Start Trial
Patent: 65584
Estimated Expiration: ⤷ Start Trial
Patent: 11521943
Estimated Expiration: ⤷ Start Trial
Patent: 14133765
Patent: METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA
Estimated Expiration: ⤷ Start Trial
Patent: 16065095
Patent: 高眼圧症及び緑内障を治療するための方法及び組成物 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 17178957
Patent: 高眼圧症及び緑内障を治療するための方法及び組成物 (METHODS AND COMPOSITIONS FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 18154656
Patent: 高眼圧症及び緑内障を治療するための方法及び組成物 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 20073574
Patent: 高眼圧症及び緑内障を治療するための方法及び組成物 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 21120412
Patent: 高眼圧症及び緑内障を治療するための方法及び組成物 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 23085558
Patent: 高眼圧症及び緑内障を治療するための方法及び組成物 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 39
Patent: طريقة ومركب لعلاج فرط ضغط العين والجلوكوما (Method and composition for treating ocular hypertension and Glaucoma)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 72249
Estimated Expiration: ⤷ Start Trial
Patent: 05334
Estimated Expiration: ⤷ Start Trial
Patent: 14877
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 9463
Patent: METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 10012987
Patent: METODO Y COMPOSICION PARA TRATAR HIPERTENSION OCULAR Y GLAUCOMA. (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA.)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 06977
Estimated Expiration: ⤷ Start Trial
Patent: 72249
Estimated Expiration: ⤷ Start Trial
Patent: 05334
Estimated Expiration: ⤷ Start Trial
Patent: 14877
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 06977
Estimated Expiration: ⤷ Start Trial
Patent: 72249
Estimated Expiration: ⤷ Start Trial
Patent: 05334
Estimated Expiration: ⤷ Start Trial
Patent: 14877
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 1628
Patent: METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 06977
Estimated Expiration: ⤷ Start Trial
Patent: 72249
Estimated Expiration: ⤷ Start Trial
Patent: 05334
Estimated Expiration: ⤷ Start Trial
Patent: 14877
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1650006
Estimated Expiration: ⤷ Start Trial
Patent: 1820816
Estimated Expiration: ⤷ Start Trial
Patent: 1988642
Estimated Expiration: ⤷ Start Trial
Patent: 2114401
Estimated Expiration: ⤷ Start Trial
Patent: 2246598
Estimated Expiration: ⤷ Start Trial
Patent: 110011707
Patent: METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA
Estimated Expiration: ⤷ Start Trial
Patent: 160102319
Patent: 고안압증과 녹내장의 치료 방법 및 조성물 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 180008905
Patent: 고안압증과 녹내장의 치료 방법 및 조성물 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 190067272
Patent: 고안압증과 녹내장의 치료 방법 및 조성물 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Patent: 200057801
Patent: 고안압증과 녹내장의 치료 방법 및 조성물 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 95316
Estimated Expiration: ⤷ Start Trial
Patent: 27837
Estimated Expiration: ⤷ Start Trial
Patent: 08050
Estimated Expiration: ⤷ Start Trial
Patent: 07982
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1000104
Patent: Method and composition for treating ocular hypertension and glaucoma
Estimated Expiration: ⤷ Start Trial
Patent: 32202
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 2257
Patent: ОФТАЛЬМОЛОГИЧЕСКИЙ ВОДНЫЙ РАСТВОР ДЛЯ ЛЕЧЕНИЯ ГЛАЗНОЙ ГИПЕРТЕНЗИИ И ГЛАУКОМЫ;ОФТАЛЬМОЛОГІЧНИЙ ВОДНИЙ РОЗЧИН ДЛЯ ЛІКУВАННЯ ОЧНОЇ ГІПЕРТЕНЗІЇ ТА ГЛАУКОМИ (OPHTHALMIC AQUEOUS COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ZIOPTAN around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Spain | 2968837 | ⤷ Start Trial | |
| European Patent Office | 2306977 | PROCÉDÉ ET COMPOSITION POUR TRAITER L'HYPERTENSION OCULAIRE ET LE GLAUCOME (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA) | ⤷ Start Trial |
| Portugal | 3205334 | ⤷ Start Trial | |
| Hungary | E049923 | ⤷ Start Trial | |
| Japan | 6356868 | ⤷ Start Trial | |
| South Korea | 20200057801 | 고안압증과 녹내장의 치료 방법 및 조성물 (METHOD AND COMPOSITION FOR TREATING OCULAR HYPERTENSION AND GLAUCOMA) | ⤷ Start Trial |
| China | 102083413 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ZIOPTAN
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0850926 | 11C0020 | France | ⤷ Start Trial | PRODUCT NAME: TAFLUPROST; REGISTRATION NO/DATE IN FRANCE: CIS:6 000 728 O DU 20110328; REGISTRATION NO/DATE AT EEC: 43230 DU 20080430 |
| 0850926 | CA 2008 00041 | Denmark | ⤷ Start Trial | |
| 0850926 | C00850926/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: TAFLUPROST; REGISTRATION NO/DATE: SWISSMEDIC 60548 27.01.2010 |
| 0850926 | SPC/GB09/005 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: TAFLUPROST OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: DK 43230 20080430; UK PL 16058/0011-0001 20081017 |
| 0850926 | 339 | Finland | ⤷ Start Trial | |
| 0850926 | SPC013/2011 | Ireland | ⤷ Start Trial | SPC013/2011: 20110808, EXPIRES: 20221221 |
| 0850926 | 91943 | Luxembourg | ⤷ Start Trial | 91943, EXPIRES: 20221222 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
ZIOPTAN: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.