Xeljanz Drug Patent Profile
✉ Email this page to a colleague
When do Xeljanz patents expire, and when can generic versions of Xeljanz launch?
Xeljanz is a drug marketed by Pfizer and Pf Prism Cv and is included in three NDAs. There are four patents protecting this drug and three Paragraph IV challenges.
This drug has seventy-five patent family members in fifty-two countries.
The generic ingredient in XELJANZ is tofacitinib citrate. There are twelve drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the tofacitinib citrate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Xeljanz
A generic version of Xeljanz was approved as tofacitinib citrate by SLAYBACK PHARMA LLC on September 25th, 2023.
Summary for Xeljanz
| International Patents: | 75 |
| US Patents: | 1 |
| Applicants: | 2 |
| NDAs: | 3 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 90 |
| Clinical Trials: | 29 |
| Patent Applications: | 106 |
| Formulation / Manufacturing: | see details |
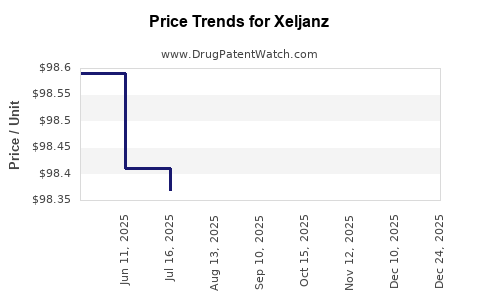
| Drug Prices: | Drug price information for Xeljanz |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for Xeljanz |
| What excipients (inactive ingredients) are in Xeljanz? | Xeljanz excipients list |
| DailyMed Link: | Xeljanz at DailyMed |
Recent Clinical Trials for Xeljanz
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Children's Hospital Los Angeles | Phase 2 |
| Fundación de Investigación Biomédica - Hospital Universitario de La Princesa | Phase 4 |
| Eli Lilly and Company | Phase 4 |
Pharmacology for Xeljanz
| Drug Class | Janus Kinase Inhibitor |
| Mechanism of Action | Janus Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for Xeljanz
Paragraph IV (Patent) Challenges for XELJANZ
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| XELJANZ | Oral Solution | tofacitinib citrate | 1 mg/mL | 213082 | 1 | 2021-11-12 |
| XELJANZ | Tablets | tofacitinib citrate | 10 mg | 203214 | 1 | 2019-07-24 |
| XELJANZ | Tablets | tofacitinib citrate | 5 mg | 203214 | 3 | 2016-11-07 |
US Patents and Regulatory Information for Xeljanz
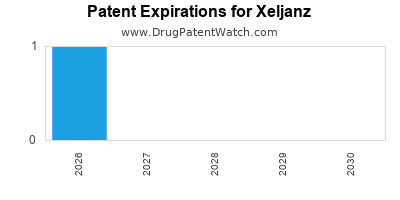
Xeljanz is protected by one US patents and one FDA Regulatory Exclusivity.
Patents protecting Xeljanz
Pyrrolo[2,3-D]pyrimidine compounds
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting Xeljanz
TREATMENT OF ADULT PATIENTS WITH ACTIVE ANKYLOSING SPONDYLITIS WHO HAVE HAD AN INADEQUATE RESPONSE OR INTOLERANCE TO ONE OR MORE TNF BLOCKERS, TO THE PRESCRIBING INFORMATION
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pfizer | XELJANZ | tofacitinib citrate | SOLUTION;ORAL | 213082-001 | Sep 25, 2020 | AA | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Pfizer | XELJANZ XR | tofacitinib citrate | TABLET, EXTENDED RELEASE;ORAL | 208246-002 | Dec 12, 2019 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-002 | May 30, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Pfizer | XELJANZ XR | tofacitinib citrate | TABLET, EXTENDED RELEASE;ORAL | 208246-001 | Feb 23, 2016 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-001 | Nov 6, 2012 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-001 | Nov 6, 2012 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Xeljanz
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-001 | Nov 6, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-001 | Nov 6, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pfizer | XELJANZ | tofacitinib citrate | SOLUTION;ORAL | 213082-001 | Sep 25, 2020 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-001 | Nov 6, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pfizer | XELJANZ | tofacitinib citrate | SOLUTION;ORAL | 213082-001 | Sep 25, 2020 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pf Prism Cv | XELJANZ | tofacitinib citrate | TABLET;ORAL | 203214-002 | May 30, 2018 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Xeljanz
See the table below for patents covering Xeljanz around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Argentina | 037635 | FORMAS CRISTALINAS Y AMORFAS DE LA SAL MONOCITRATO DE 3-[4-METIL-3-[METIL-(7H-PIRROLO[2,3-D] PIRIMIDIN-4-IL)-AMINO]- PIPERIDIN-1-IL)-3-OXO-PROPIONITRILO, COMPOSICIONES FARMACEUTICAS QUE LO CONTIENEN, USO EN LA MANUFACTURA DE MEDICAMENTOS Y PROCEDIMIENTO PARA OBTENER UNA FORMA CRISTALIZADA | ⤷ Try a Trial |
| New Zealand | 540332 | Optical resolution of (1-benzyl-4-methylpiperidin-3-YL) - methylamine and the use thereof for the preparation of pyrrolo 2,3-pyrimidine derivatives as protein kinases inhibitors | ⤷ Try a Trial |
| Denmark | 1666481 | ⤷ Try a Trial | |
| Mexico | PA03011062 | RESOLUCION OPTICA DE (1-BENCIL-4-METILPIPERIDIN-3-IL)-METILAMINA Y EL USO DE LA MISMA PARA LA PREPARACION DE DERIVADOS DE PIRROLO 2,3-PIRIMIDINA COMO INHIBIDORES DE PROTEINA QUINASAS. (OPTICAL RESOLUTION OF (1-BENZYL-4-METHYLPIPERIDIN-3-YL) -METHYLAMINE AND THE USE THEREOF FOR THE PREPARATION OF PYRROLO 2,3-PYRIMIDINE DERIVATIVES AS PROTEIN KINASES INHIBITORS.) | ⤷ Try a Trial |
| Estonia | 05332 | 3-{(3R,4R)-4-met l-3-[met l(7H-p rrolo[2,3-d]p rimidiin-4- l)amino]piperidiin-1 - l}-3-oksopropionitriil, selle farmatseutiliselt vastuv?etav sool ja nende kasutamine | ⤷ Try a Trial |
| South Africa | 200307982 | Optical resolution of (1-benzyl-4-methylpiperidin-3-yl)-methylamine and the use thereof for the preparation of pyrrolo 2,3-pyrimidine derivatives as protein kinases inhibitors. | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for Xeljanz
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1666481 | 2017/036 | Ireland | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, INCLUDING THE CITRATE SALT; REGISTRATION NO/DATE: EU/1/17/1178/001 EU/1/17/1178/004 20170322 |
| 1666481 | 122017000062 | Germany | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB ODER EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/17/1178 20170322 |
| 1666481 | PA2017025 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIBAS, PASIRINKTINAI FARMACINIU POZIURIU PRIIMTINOS DRUSKOS PAVIDALE, ISKAITANT CITRATO DRUSKAAA; REGISTRATION NO/DATE: EU/1/17/1178 20170322 |
| 1666481 | C201730033 | Spain | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB Y SALES FARMACEUTICAMENTE ACEPTABLES DEL MISMO, INCLUYENDO LA SAL CITRATO; NATIONAL AUTHORISATION NUMBER: EU/1/17/1178; DATE OF AUTHORISATION: 20170322; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/17/1178; DATE OF FIRST AUTHORISATION IN EEA: 20170322 |
| 1666481 | LUC00031 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB, EVENTUELLEMENT SOUS LA FORME D'UN SEL PHARMACEUTIQUEMENT ACCEPTABLE, Y COMPRIS LE SEL DE CITRATE; AUTHORISATION NUMBER AND DATE: EU/1/17/1178 20170324 |
| 1666481 | C 2017 029 | Romania | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB, OPTIONAL SUB FORMA UNEI SARI ACCEPTABILE FARMACEUTIC, INCLUSIV SAREA CITRAT 3-((3R,4R)-4-METIL-3-{METIL-(7H-PIROLO[2,3-D]PIRIMIDIN-4-IL)-AMINO]PERIDINIL)-3-OXO-PROPIONITRIL; NATIONAL AUTHORISATION NUMBER: EU/1/17/1178; DATE OF NATIONAL AUTHORISATION: 20170322; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/17/1178; DATE OF FIRST AUTHORISATION IN EEA: 20170322 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


