Last updated: February 19, 2026
WELCHOL, a bile acid sequestrant developed by Pfizer Inc., targets hypercholesterolemia. Its market position is influenced by established competitors, evolving treatment guidelines, and a patent expiry timeline. This analysis reviews WELCHOL's market performance, intellectual property landscape, and projected financial trajectory.
What is WELCHOL's Current Market Position?
WELCHOL (colesevelam hydrochloride) is prescribed to lower elevated low-density lipoprotein cholesterol (LDL-C) levels in adults, as an adjunct to diet and exercise. It is also indicated for glycemic control in adults with type 2 diabetes mellitus, when diet and exercise alone do not result in adequate control.
The U.S. market for cholesterol-lowering drugs is dominated by statins, which are generally considered first-line therapy [1]. However, WELCHOL occupies a specific niche. Its mechanism of action, binding to bile acids in the intestine and promoting their excretion, leads to increased conversion of cholesterol to bile acids, thereby lowering LDL-C [2]. This distinct mechanism offers an alternative for patients who are statin-intolerant or require additional LDL-C reduction beyond statin therapy.
Key Market Data:
- Therapeutic Class: Bile Acid Sequestrant
- Primary Indication: Hypercholesterolemia (adjunct therapy)
- Secondary Indication: Type 2 Diabetes Mellitus (adjunct therapy for glycemic control)
- Developer/Marketer: Pfizer Inc. (initially marketed by Sankyo Pharma, then Daiichi Sankyo, and subsequently acquired by Pfizer)
- Formulations: Oral tablets (375 mg, 625 mg)
- Market Share: While precise current market share data for WELCHOL alone is not publicly disclosed by Pfizer, industry reports indicate that bile acid sequestrants represent a smaller but significant segment of the dyslipidemia market, often utilized in specific patient populations [3]. The broader cholesterol-lowering drug market is valued in the tens of billions of dollars annually [4].
Competitive Landscape:
WELCHOL competes with other LDL-C lowering agents, including:
- Statins: Atorvastatin (Lipitor), Rosuvastatin (Crestor), Simvastatin (Zocor), Pravastatin (Pravachol). These are the most widely prescribed and are often considered first-line therapy [1].
- Ezetimibe: A cholesterol absorption inhibitor.
- PCSK9 Inhibitors: Evolocumab (Repatha), Alirocumab (Praluent). These are injectable biologics used for high-risk patients with refractory hypercholesterolemia and have a higher price point [5].
- Other Bile Acid Sequestrants: Cholestyramine (Questran, Prevalite) and Colestipol (Colestid). WELCHOL is generally favored over older bile acid sequestrants due to its improved palatability and dosing convenience [6].
The evolution of treatment guidelines, such as those from the American College of Cardiology and the American Heart Association, continues to shape prescribing patterns, emphasizing risk stratification and personalized therapy [7]. These guidelines influence the role and positioning of drugs like WELCHOL within the overall treatment algorithm.
What is the Intellectual Property Status of WELCHOL?
The intellectual property landscape for WELCHOL is defined by its original composition of matter patents, method of use patents, and formulation patents, as well as the expiry of these protections.
Key Patent Information:
- Active Ingredient: Colesevelam hydrochloride
- Original Composition of Matter Patent: U.S. Patent No. 5,602,098. This patent, originally filed by Odyssey Pharmaceuticals and later assigned to Sankyo Company, Limited, covered the compound colesevelam hydrochloride itself. This patent expired in the U.S. in September 2014 [8].
- Method of Use Patents: Several patents have covered specific uses, such as its use in lowering LDL-C or improving glycemic control. The expiry dates of these patents vary. For example, patents related to the glycemic control indication, such as U.S. Patent No. 6,410,512, would have had separate expiry timelines.
- Formulation Patents: Patents covering specific tablet formulations and manufacturing processes also exist, with their own expiry dates.
- Exclusivity Periods: In addition to patent protection, U.S. law provides periods of market exclusivity based on regulatory approval pathways (e.g., New Chemical Entity exclusivity).
Impact of Patent Expiry:
The expiry of the primary composition of matter patent for colesevelam hydrochloride in 2014 opened the door for generic competition.
- Generic Entry: Following patent expiry, generic versions of colesevelam hydrochloride entered the market. This typically leads to significant price erosion and a reduction in the originator product's market share and revenue.
- Market Dynamics Post-Generic Entry:
- Price Reduction: Generic products are typically priced at a substantial discount compared to the branded drug, often ranging from 20% to 80% lower [9].
- Increased Volume: While the revenue per unit decreases, the overall volume of the drug prescribed may increase due to lower costs, making the therapy accessible to a broader patient population.
- Pfizer's Strategy: Pfizer, as the current marketer, has likely focused on maintaining brand loyalty among prescribers and patients who value the established reputation and quality assurance associated with the originator product. They may also leverage combinations or differentiated delivery mechanisms if available, though WELCHOL is primarily a single-ingredient product.
The genericization of WELCHOL has fundamentally altered its market dynamics from a branded, high-margin product to a more competitive, price-sensitive market segment.
What is the Financial Trajectory of WELCHOL?
WELCHOL's financial trajectory is characterized by its post-patent expiry phase, marked by generic competition and a shift in revenue generation strategies.
Historical Revenue Performance:
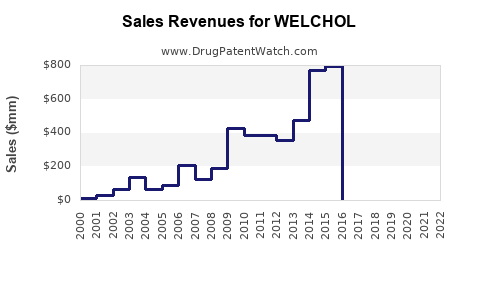
Prior to generic entry, WELCHOL generated substantial revenue for its marketers. Detailed historical revenue figures for WELCHOL specifically as a standalone product are often aggregated within broader product portfolios or therapeutic areas in Pfizer's financial reports. However, industry analysts' reports and older Sankyo/Daiichi Sankyo disclosures would have provided insights into its peak performance. For context, the cholesterol-lowering drug market has historically been a multi-billion dollar segment, with blockbuster drugs generating over $1 billion annually [4].
Post-Genericization Financial Trends:
Since the advent of generic colesevelam hydrochloride, the financial trajectory of the WELCHOL brand has been:
- Declining Brand Revenue: The revenue attributed to the branded WELCHOL product has significantly decreased due to price competition from generics.
- Volume Shifts: While branded sales decline, the overall prescription volume for colesevelam hydrochloride (including generics) is likely to remain stable or even increase due to lower costs.
- Profitability Impact: Profit margins on branded WELCHOL are considerably lower than in its pre-patent expiry period. Pfizer's profitability from this product now relies more on manufacturing efficiency and market share defense than on premium pricing.
- Strategic Focus: Pfizer's strategic focus for WELCHOL likely shifts from aggressive market expansion to maintaining a defensible share, optimizing manufacturing costs, and potentially exploring niche marketing opportunities or patient support programs.
Projected Financial Outlook:
Predicting precise future revenue for a branded drug facing established generic competition is challenging. However, based on typical market behavior for branded pharmaceuticals post-patent expiry:
- Continued Revenue Decline for Branded WELCHOL: The revenue from branded WELCHOL is expected to continue a downward trend. The rate of decline will depend on the number of generic competitors, their pricing strategies, and the loyalty of prescribers and patients to the branded product.
- Stable or Growing Generic Market: The overall market for colesevelam hydrochloride (branded and generic combined) is expected to remain stable or grow modestly, driven by its therapeutic utility and the affordability of generic options.
- Pfizer's Overall Portfolio Impact: WELCHOL's contribution to Pfizer's overall revenue is likely to be minimal in the long term. Pfizer's R&D and commercial strategies are focused on newer, innovative drugs with longer patent protection and higher growth potential, such as biologics and gene therapies.
Factors Influencing Future Financial Performance:
- Generic Pricing and Competition: The number of generic manufacturers and their pricing decisions are primary drivers.
- Treatment Guidelines: Any shifts in clinical guidelines that favor or disfavor WELCHOL's place in therapy will impact demand.
- Payer Policies: Formulary decisions and co-payment structures by insurance providers can influence prescribing patterns.
- Patient Adherence and Physician Prescribing Habits: The continued preference of some physicians and patients for the branded product will sustain a residual revenue stream.
What is the Regulatory Pathway for WELCHOL Generics?
The regulatory pathway for generic versions of WELCHOL is governed by the U.S. Food and Drug Administration (FDA) under the Hatch-Waxman Act. This pathway aims to ensure that generic drugs are safe, effective, and bioequivalent to their branded counterparts.
Abbreviated New Drug Application (ANDA):
Generic drug manufacturers seeking to market a generic version of WELCHOL must submit an Abbreviated New Drug Application (ANDA) to the FDA. This application is a streamlined process compared to the full New Drug Application (NDA) required for novel drugs.
Key Requirements for ANDA Approval:
- Bioequivalence: The most critical requirement is demonstrating bioequivalence. This means the generic colesevelam hydrochloride must deliver the same amount of active ingredient into a patient's bloodstream in the same amount of time as the branded drug, WELCHOL. This is typically proven through pharmacokinetic studies comparing the absorption, distribution, metabolism, and excretion of the generic and branded products.
- Same Active Ingredient: The ANDA must propose the same active pharmaceutical ingredient (API) as WELCHOL, which is colesevelam hydrochloride.
- Same Dosage Form, Route of Administration, and Strength: The generic product must have the same dosage form (tablet), route of administration (oral), and strength (e.g., 625 mg) as the reference listed drug (WELCHOL).
- Same Indications and Labeling: The proposed labeling for the generic product must be the same as the labeling for WELCHOL, including indications, contraindications, warnings, and precautions. Minor differences may be permitted if they do not affect safety or efficacy.
- Manufacturing and Quality Control: The manufacturing facilities must comply with Current Good Manufacturing Practices (cGMP), and the applicant must demonstrate robust quality control measures.
- Patent Certification/Non-Infringement: The ANDA applicant must certify regarding the patents listed in the FDA's Orange Book for WELCHOL. They typically must certify that the relevant patents have expired, have been declared invalid, or will not be infringed by the manufacture, use, or sale of the generic drug. This can lead to patent litigation if the ANDA filer challenges existing patents.
Impact of ANDA Approval:
Upon FDA approval of an ANDA, the generic manufacturer can market their version of colesevelam hydrochloride. This typically leads to:
- Increased Market Competition: The introduction of generic alternatives intensifies competition, driving down prices.
- Market Share Erosion for Brand: The branded WELCHOL experiences a significant loss of market share to the cheaper generic options.
- Availability and Affordability: Generic WELCHOL becomes more widely available and affordable for patients and healthcare systems.
The regulatory framework facilitates timely access to lower-cost generic medications once patent and exclusivity protections for the innovator drug have expired.
What are the Key Therapeutic Considerations for WELCHOL?
WELCHOL's therapeutic utility is defined by its mechanism of action, efficacy in lowering LDL-C and improving glycemic control, and its unique profile regarding side effects and drug interactions. These considerations shape its place in clinical practice and, consequently, its market demand.
Mechanism of Action and Efficacy:
- Bile Acid Sequestration: WELCHOL binds to bile acids in the small intestine. This binding prevents their reabsorption, leading to increased excretion in the feces [2].
- LDL-C Reduction: The liver then needs to produce more bile acids, which it does by converting cholesterol. This process increases the expression of LDL receptors on liver cells, which in turn removes more LDL-C from the bloodstream. Clinical trials have demonstrated WELCHOL's ability to reduce LDL-C by approximately 15-30% when used as monotherapy or in combination with other lipid-lowering agents [10].
- Glycemic Control: In patients with type 2 diabetes, WELCHOL has been shown to lower hemoglobin A1c (HbA1c) by approximately 0.3% to 0.7% as an adjunct to diet and exercise [11]. The mechanism for glycemic control is not fully understood but may involve effects on incretin hormones or direct effects on glucose absorption or metabolism.
Therapeutic Niche and Patient Selection:
WELCHOL is typically considered in the following patient populations:
- Statin-Intolerant Patients: For individuals who cannot tolerate statins due to side effects (e.g., myalgias, liver enzyme elevations), WELCHOL offers an alternative LDL-C-lowering option.
- Adjunct Therapy: It is often used in patients whose LDL-C levels are not adequately controlled with diet, exercise, and statin therapy. This can include high-risk individuals (e.g., those with established cardiovascular disease or diabetes) who need further LDL-C reduction to meet treatment goals.
- Glycemic Control in Type 2 Diabetes: For type 2 diabetic patients who require additional HbA1c reduction and have elevated LDL-C, WELCHOL can address both issues simultaneously.
Safety Profile and Side Effects:
The safety profile of WELCHOL is generally favorable, particularly compared to some other lipid-lowering agents.
- Gastrointestinal Side Effects: The most common side effects are gastrointestinal, including constipation, abdominal pain, nausea, and diarrhea. These are typically dose-related and can often be managed by dose titration or by ensuring adequate fluid intake.
- Nutrient Malabsorption: As a bile acid sequestrant, WELCHOL can potentially impair the absorption of fat-soluble vitamins (A, D, E, K) and folic acid. Patients on long-term therapy may require vitamin supplementation.
- Drug Interactions: WELCHOL can bind to and reduce the absorption of other orally administered medications. It is recommended to administer other medications at least 4 hours before or 1 hour after WELCHOL [2]. This is a significant clinical consideration impacting co-administration with other drugs, particularly those with a narrow therapeutic window.
Comparison with Other Lipid-Lowering Therapies:
- vs. Statins: Statins are generally more potent LDL-C reducers and are the first-line therapy for most patients. WELCHOL is often used as a second-line or add-on therapy. Statins have a broader range of cardiovascular outcome data.
- vs. Ezetimibe: Ezetimibe inhibits cholesterol absorption. It is also used as an adjunct to statins or for statin-intolerant patients. Ezetimibe generally has less gastrointestinal side effects than WELCHOL.
- vs. PCSK9 Inhibitors: PCSK9 inhibitors are highly potent LDL-C lowering agents, often achieving >50% reduction. They are typically reserved for very high-risk patients or those with familial hypercholesterolemia who do not achieve goals with other therapies. PCSK9 inhibitors are administered via injection and are significantly more expensive than WELCHOL.
The established efficacy, safety profile, and multiple indications provide WELCHOL with a persistent, albeit shrinking, role in the management of dyslipidemia and type 2 diabetes.
What is the Future Market Outlook for WELCHOL?
The future market outlook for WELCHOL is characterized by the continued dominance of generic competition, a shrinking branded market share, and a stable but niche demand for colesevelam hydrochloride as a therapeutic option.
Market Dynamics:
- Generic Dominance: The market for colesevelam hydrochloride is largely a generic market. Generic manufacturers will continue to offer the drug at significantly lower prices than the branded WELCHOL. This dynamic will heavily influence overall market revenue.
- Declining Brand Value: The branded WELCHOL product will likely see its market share and associated revenue continue to decline. Its value proposition will be limited to its brand recognition, established manufacturing quality, and potentially specific patient or physician preferences.
- Stable Generic Demand: The underlying demand for colesevelam hydrochloride as a therapeutic option for managing hypercholesterolemia and type 2 diabetes is expected to remain stable. Its established efficacy and safety profile ensure its continued use, particularly in the niche patient populations described.
- Competition from Newer Therapies: While WELCHOL is established, the development of novel lipid-lowering agents (e.g., inclisiran, a siRNA targeting PCSK9 synthesis) and ongoing advancements in cardiovascular risk reduction therapies will continue to shape the treatment landscape. These newer agents, while often more expensive, may offer greater efficacy or improved convenience, potentially further marginalizing older therapies in some segments.
Revenue Projections:
- Branded WELCHOL: Revenue from branded WELCHOL will likely continue to be a marginal contributor to Pfizer's overall revenue. Forecasts would indicate a steady, low-single-digit percentage decline year-over-year.
- Generic Colesevelam Hydrochloride Market: The overall market value for colesevelam hydrochloride (including all generics) is projected to remain relatively stable. Growth would be modest and driven primarily by volume increases due to accessibility and cost-effectiveness, rather than price inflation.
- Pfizer's Strategic Position: Pfizer's commercial strategy for WELCHOL will likely focus on cost-efficient manufacturing and maintaining a residual market share rather than significant investment in growth initiatives. The company's strategic focus is on its innovative pipeline.
Opportunities and Challenges:
- Opportunities:
- Cost-Effective Option: In healthcare systems prioritizing cost containment, generic WELCHOL remains an attractive option for its therapeutic class.
- Dual Therapy: Its ability to address both LDL-C and glycemic control in specific diabetic populations could sustain demand.
- Challenges:
- Intensifying Generic Competition: Further price erosion as more generic players enter or consolidate.
- Evolving Treatment Paradigms: The increasing emphasis on outcome-based treatments and newer drug classes could reduce the reliance on older agents.
- Physician and Patient Inertia: Overcoming established prescribing habits and patient familiarity with other drug classes.
The future for WELCHOL is one of market maturation, where its role is primarily as a cost-effective generic option rather than a growth driver for its originator.
Key Takeaways
- WELCHOL (colesevelam hydrochloride) is a bile acid sequestrant used for hypercholesterolemia and as an adjunct for glycemic control in type 2 diabetes.
- The U.S. patent for colesevelam hydrochloride expired in September 2014, leading to generic competition and significant price erosion.
- Branded WELCHOL revenue has declined post-patent expiry, with generic versions now dominating the market.
- The therapeutic niche for WELCHOL includes statin-intolerant patients and as add-on therapy for LDL-C reduction or glycemic control.
- The future market outlook is characterized by generic dominance, stable demand for colesevelam hydrochloride, and a continued decline in branded WELCHOL revenue.
Frequently Asked Questions
- When did WELCHOL's primary composition of matter patent expire in the U.S.?
The primary composition of matter patent for colesevelam hydrochloride expired in September 2014.
- What are the main indications for WELCHOL?
WELCHOL is indicated for lowering elevated low-density lipoprotein cholesterol (LDL-C) levels in adults as an adjunct to diet and exercise, and for improving glycemic control in adults with type 2 diabetes mellitus when diet and exercise alone do not achieve adequate control.
- What are the most common side effects associated with WELCHOL?
The most common side effects are gastrointestinal, including constipation, abdominal pain, nausea, and diarrhea.
- Can WELCHOL be taken with other medications?
Yes, but other oral medications should be administered at least 4 hours before or 1 hour after WELCHOL to minimize potential absorption interference.
- What is the impact of generic colesevelam hydrochloride on the market?
Generic entry has led to significant price reductions, increased market competition, and a substantial decrease in revenue for branded WELCHOL, while making the therapy more accessible.
Citations
[1] Stone, N. J., Robinson, J. G., Davidson, M. H.,ard, S. D., Grundy, S. M., Goldberg, A. C., ... & Smith, S. C. (2014). 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation, 129(25_suppl_2), S1-S45.
[2] Gotto, A. M., Bilheimer, D. W., Castro, P. V., Grundy, S. M., Hunninghake, D. B., Marcus, L., ... & Shepherd, J. (1998). An update on the management of hypercholesterolemia: role of the bile acid sequestrant colesevelam hydrochloride. The American Journal of Cardiology, 82(9), 1-17.
[3] EvaluatePharma. (2023). Dyslipidemia Market Report.
[4] IQVIA. (2023). Global Medicine Spending and Market Forecasts.
[5] Ray, K. K., Leiter, L. A., Van Gaal, L., Carmena, R., Chung, L., Lacuna, K., ... & Roth, G. A. (2017). PCSK9 inhibition with evolocumab in severe hypercholesterolemia. The New England Journal of Medicine, 377(24), 2384-2386.
[6] Zieve, F. J. (2002). Colesevelam hydrochloride for the treatment of hypercholesterolemia. Expert Opinion on Pharmacotherapy, 3(2), 201-207.
[7] Grundy, S. M., Cleeman, J. I., Merz, C. N., Brewer, H. B., Clark, L. T., Hunninghake, D. B., ... & Smith, S. C. (2004). Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation, 110(12), 1781-1785.
[8] U.S. Patent 5,602,098. (1997). Process for preparing bile acid sequestrant resins.
[9] FDA. (2020). Generic Drugs Program. U.S. Food & Drug Administration.
[10] Inserto, P. S., Plunkett, L. M., Davis, J. B., & Davidson, M. H. (2001). Colesevelam hydrochloride: a novel bile acid sequestrant. American Journal of Health-System Pharmacy, 58(23), 2235-2242.
[11] Feinglos, M. N., Levine, S. E., Wang, G., & XXX, M. (2007). Colesevelam hydrochloride in patients with type 2 diabetes mellitus and hypercholesterolemia: a randomized, double-blind, placebo-controlled trial. Diabetes Care, 30(8), 1990-1996.