Last updated: February 18, 2026
This report analyzes the market dynamics and financial trajectory of Valtrex (valacyclovir hydrochloride), a prodrug of acyclovir used to treat herpes simplex virus (HSV) infections and varicella zoster virus (VZV) infections, including shingles and chickenpox. The analysis covers patent expiries, generic competition, market size, and projected financial performance.
What is Valtrex and Its Mechanism of Action?
Valtrex is an antiviral medication. It functions as a prodrug, meaning it is converted in the body into its active form, acyclovir. Acyclovir selectively inhibits the replication of herpesviruses. Specifically, it acts as a substrate for viral thymidine kinase, an enzyme present in HSV and VZV infected cells but not in healthy cells. This viral enzyme phosphorylates acyclovir to acyclovir monophosphate. Cellular kinases then further phosphorylate it to acyclovir triphosphate, which is the active antiviral agent. Acyclovir triphosphate inhibits viral DNA polymerase and is incorporated into viral DNA, leading to chain termination. This mechanism effectively halts viral replication [1].
Valtrex is indicated for the treatment of:
- Genital herpes: To cure initial outbreaks and reduce the frequency and severity of recurrent episodes [1].
- Cold sores (herpes labialis): To speed healing and reduce the number of outbreaks [1].
- Shingles (herpes zoster): To reduce pain and duration of illness [1].
- Chickenpox (varicella): To reduce the duration and severity of symptoms [1].
What is the Patent and Exclusivity Status of Valtrex?
The original patent for valacyclovir hydrochloride was filed by Glaxo Group Limited (now GlaxoSmithKline, GSK) and granted in the United States as U.S. Patent No. 4,352,812. The patent was issued on October 5, 1982, with an expiration date of October 5, 2002. However, extensions and other forms of exclusivity often apply.
Key patent and exclusivity milestones for Valtrex include:
- Original Expiry: U.S. Patent No. 4,352,812 expired on October 5, 2002 [2].
- Pediatric Exclusivity: In 2001, GSK received six months of pediatric exclusivity for Valtrex, extending market exclusivity until April 5, 2002 [3].
- ANDA Filings and Litigation: Following the expiry of its primary patent and exclusivity, Valtrex faced significant challenges from generic manufacturers seeking U.S. Food and Drug Administration (FDA) approval via Abbreviated New Drug Applications (ANDAs). This led to extensive patent litigation, a common scenario for blockbuster drugs nearing patent expiry. GlaxoSmithKline defended its patent rights aggressively, but numerous generic versions eventually entered the market.
- Loss of Exclusivity Date: The effective loss of market exclusivity, allowing for widespread generic entry in the U.S., occurred in 2009 after significant patent challenges and litigation surrounding secondary patents [4].
- International Patent Status: Patent protection varies by country. While the primary composition of matter patents may have expired in many regions, other patents related to formulations, methods of use, or manufacturing processes could extend protection or lead to legal disputes. For instance, in Europe, the Supplementary Protection Certificate (SPC) system provides extensions to patent terms.
What is the Market Size and Revenue for Valtrex?
Valtrex was a significant revenue generator for GlaxoSmithKline (GSK) during its period of market exclusivity.
Historical Revenue Data:
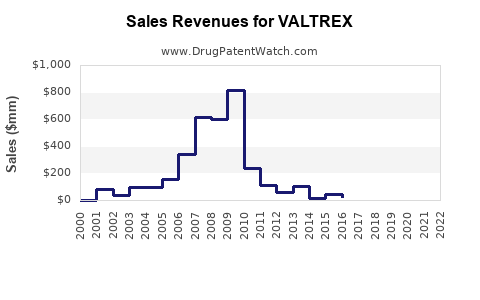
- Peak Sales: Valtrex achieved peak annual sales of over $3 billion globally in the mid-2000s, prior to widespread generic competition [5].
- U.S. Sales: In the U.S., sales of Valtrex before generic entry were substantial, contributing significantly to GSK's antiviral portfolio. For example, in 2008, U.S. sales were approximately $1.5 billion [6].
- Global Sales Trend:
- 2006: $2.3 billion [7]
- 2007: $2.4 billion [7]
- 2008: $2.4 billion [6]
- 2009: $2.1 billion (pre-generic impact significant) [8]
- 2010: $1.3 billion (significant impact from generic competition) [8]
- 2011: $880 million [8]
- 2012: $650 million [9]
The sharp decline in revenue from 2009 onwards is a direct consequence of generic manufacturers entering the market with significantly lower-priced alternatives.
How Has Generic Competition Impacted Valtrex Sales?
The introduction of generic valacyclovir hydrochloride has fundamentally altered the market dynamics for the drug.
- Price Erosion: Generic versions of valacyclovir are priced substantially lower than branded Valtrex. This price difference typically ranges from 30% to 70%, depending on the market and supplier.
- Market Share Shift: Branded Valtrex has lost the vast majority of its market share to generics. Prescriptions are now predominantly filled with generic valacyclovir due to cost savings for patients and healthcare systems.
- Volume vs. Value: While the total volume of valacyclovir prescribed (branded and generic combined) may remain relatively stable or even increase due to accessibility, the overall market value for the active pharmaceutical ingredient (API) has decreased dramatically.
- Continued Sales for GSK (Reduced): GSK continues to sell Valtrex, but its revenue from the drug is now a fraction of its former peak. This revenue comes from a smaller segment of the market that may still prefer the branded product for various reasons (e.g., physician preference, specific patient formulary access, although this is increasingly rare).
- Generic Market Growth: The market for generic valacyclovir has expanded significantly, with multiple manufacturers competing. This competition further drives down prices and ensures broad availability. Major generic players often include companies like Teva Pharmaceutical Industries, Mylan (now Viatris), and generic divisions of larger pharmaceutical companies.
What are the Current Market Trends and Future Outlook?
The market for valacyclovir is now mature and dominated by generic competition.
- Stable but Low Revenue: For branded Valtrex, future revenue streams are projected to remain low, reflecting its niche position against generics. GSK's strategy likely focuses on managing the remaining profitability rather than significant growth initiatives for the branded product.
- Generic Market Dynamics: The generic valacyclovir market is characterized by intense price competition and volume-driven sales. Profitability for generic manufacturers is dependent on efficient production, supply chain management, and securing favorable contracts with wholesalers and pharmacy benefit managers (PBMs).
- Therapeutic Landscape: While newer antiviral agents may emerge for specific indications or resistant strains, valacyclovir remains a first-line or cost-effective option for common HSV and VZV infections. Its established safety profile and efficacy contribute to its continued use.
- Geographic Variations: The pace of genericization and subsequent price erosion can vary by country due to differing regulatory pathways, patent laws, and healthcare reimbursement policies. However, most developed markets have experienced widespread generic availability for many years.
- Emerging Markets: In some emerging markets, branded Valtrex may retain a larger market share for a longer period due to slower generic entry or specific market access strategies. However, the global trend is towards generic dominance.
- Potential for New Indications (Limited): While ongoing research could theoretically identify new therapeutic uses for valacyclovir, the likelihood of developing entirely new, high-revenue indications for a long-established, off-patent drug is low. Any new uses would likely be pursued by generic manufacturers or through academic research, with limited commercial impact for the original brand.
Key Financial Trajectory Insights
The financial trajectory of Valtrex represents a classic pharmaceutical product lifecycle: rapid growth during patent-protected exclusivity, followed by a sharp decline upon generic entry, and eventual stabilization at a much lower revenue level for the branded product, with the overall API market volume continuing under generic manufacturers.
- Peak Period: Mid-2000s, characterized by high single-digit to low double-digit annual growth.
- Patent Expiry & Generic Entry: 2009-2010 marked the inflection point, leading to immediate and substantial revenue decline for the branded product.
- Post-Genericization: Revenue for branded Valtrex stabilized at a significantly reduced level, representing a small fraction of its peak sales. The overall market for valacyclovir API is now driven by the volume and competitive pricing of generic manufacturers.
Key Takeaways
- Valtrex, a prodrug of acyclovir, lost U.S. market exclusivity in 2009.
- Peak global sales for branded Valtrex exceeded $3 billion annually before generic competition.
- Generic valacyclovir entered the market significantly lowering prices and eroding branded market share.
- GSK's revenue from Valtrex has declined by over 75% from its peak.
- The current market for valacyclovir is dominated by generic manufacturers, with intense price competition.
- Branded Valtrex continues to generate minimal revenue, catering to a small market segment.
- The financial trajectory demonstrates a typical lifecycle for a blockbuster drug, from rapid growth to mature genericization.
Frequently Asked Questions
-
When did the primary U.S. patent for Valtrex expire?
The primary U.S. patent for Valtrex (U.S. Patent No. 4,352,812) expired on October 5, 2002.
-
What is the primary reason for the significant decline in Valtrex sales since 2009?
The significant decline in sales is due to the widespread entry of generic valacyclovir hydrochloride into the market after the expiration of key patents and exclusivity periods, leading to substantial price erosion.
-
Does GlaxoSmithKline still manufacture and sell Valtrex?
Yes, GlaxoSmithKline continues to manufacture and sell branded Valtrex, but its market share and revenue are now a small fraction of their historical peak due to generic competition.
-
What are the main indications for Valtrex?
Valtrex is indicated for the treatment of genital herpes, cold sores, shingles, and chickenpox, all caused by herpes simplex virus and varicella zoster virus.
-
What is the expected future revenue trend for branded Valtrex?
The revenue for branded Valtrex is expected to remain low and stable, reflecting its current niche market position against significantly cheaper generic alternatives.
Citations
[1] U.S. Food & Drug Administration. (n.d.). DailyMed - Valtrex- valacyclovir hydrochloride tablet. National Library of Medicine. Retrieved from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=7557d16c-603f-44b8-889d-2d9637220052
[2] U.S. Patent and Trademark Office. (1982). U.S. Patent 4,352,812: Substituted 2-amino-1,3-propanediols.
[3] U.S. Food & Drug Administration. (2001). Application Number: 050756. (Pediatric exclusivity granted).
[4] Generic Pharmaceutical Association. (2009). Generic Valtrex Launched. (Press Release).
[5] GlaxoSmithKline plc. (2006). Annual Report and Accounts 2006.
[6] GlaxoSmithKline plc. (2009). Annual Report and Accounts 2009.
[7] GlaxoSmithKline plc. (2008). Annual Report and Accounts 2008.
[8] GlaxoSmithKline plc. (2011). Annual Report and Accounts 2011.
[9] GlaxoSmithKline plc. (2012). Annual Report and Accounts 2012.