UPTRAVI Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Uptravi, and what generic alternatives are available?
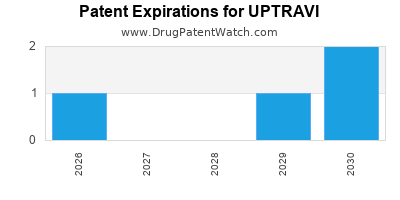
Uptravi is a drug marketed by Actelion and is included in two NDAs. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and seventy-seven patent family members in forty-two countries.
The generic ingredient in UPTRAVI is selexipag. There are two drug master file entries for this compound. Three suppliers are listed for this compound. Additional details are available on the selexipag profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Uptravi
A generic version of UPTRAVI was approved as selexipag by ALEMBIC on October 11th, 2023.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for UPTRAVI?
- What are the global sales for UPTRAVI?
- What is Average Wholesale Price for UPTRAVI?
Summary for UPTRAVI
| International Patents: | 177 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 78 |
| Clinical Trials: | 5 |
| Patent Applications: | 466 |
| Drug Prices: | Drug price information for UPTRAVI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for UPTRAVI |
| What excipients (inactive ingredients) are in UPTRAVI? | UPTRAVI excipients list |
| DailyMed Link: | UPTRAVI at DailyMed |
Recent Clinical Trials for UPTRAVI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Humanis Saglk Anonim Sirketi | PHASE1 |
| University of Sheffield | Phase 4 |
| University of Cambridge | Phase 4 |
Pharmacology for UPTRAVI
| Drug Class | Prostacyclin Receptor Agonist |
| Mechanism of Action | Prostacyclin Receptor Agonists |
US Patents and Regulatory Information for UPTRAVI
UPTRAVI is protected by six US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Actelion | UPTRAVI | selexipag | TABLET;ORAL | 207947-007 | Dec 21, 2015 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Actelion | UPTRAVI | selexipag | TABLET;ORAL | 207947-005 | Dec 21, 2015 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Actelion | UPTRAVI | selexipag | TABLET;ORAL | 207947-008 | Dec 21, 2015 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for UPTRAVI
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Janssen Cilag International NV | Uptravi | selexipag | EMEA/H/C/003774Uptravi is indicated for the long-term treatment of pulmonary arterial hypertension (PAH) in adult patients with WHO functional class (FC) II–III, either as combination therapy in patients insufficiently controlled with an endothelin receptor antagonist (ERA) and/or a phosphodiesterase type 5 (PDE-5) inhibitor, or as monotherapy in patients who are not candidates for these therapies., , Efficacy has been shown in a PAH population including idiopathic and heritable PAH, PAH associated with connective tissue disorders, and PAH associated with corrected simple congenital heart disease., | Authorised | no | no | no | 2016-05-12 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for UPTRAVI
When does loss-of-exclusivity occur for UPTRAVI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 7242
Estimated Expiration: ⤷ Start Trial
Patent: 1658
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10263569
Estimated Expiration: ⤷ Start Trial
Patent: 16366073
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2018009534
Estimated Expiration: ⤷ Start Trial
Patent: 2021005510
Estimated Expiration: ⤷ Start Trial
Patent: 1015936
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 64475
Estimated Expiration: ⤷ Start Trial
Patent: 05169
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11003264
Estimated Expiration: ⤷ Start Trial
Patent: 18001464
Estimated Expiration: ⤷ Start Trial
China
Patent: 2459198
Estimated Expiration: ⤷ Start Trial
Patent: 4326991
Estimated Expiration: ⤷ Start Trial
Patent: 8289890
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 30432
Estimated Expiration: ⤷ Start Trial
Patent: 18006834
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0180171
Estimated Expiration: ⤷ Start Trial
Patent: 0200539
Estimated Expiration: ⤷ Start Trial
Patent: 0250572
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 19788
Estimated Expiration: ⤷ Start Trial
Patent: 22893
Estimated Expiration: ⤷ Start Trial
Patent: 18011
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Patent: 75871
Estimated Expiration: ⤷ Start Trial
Patent: 31607
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 18049108
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Patent: 75871
Estimated Expiration: ⤷ Start Trial
Patent: 84911
Estimated Expiration: ⤷ Start Trial
Patent: 89855
Estimated Expiration: ⤷ Start Trial
Patent: 31607
Estimated Expiration: ⤷ Start Trial
Patent: 01404
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 31607
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 44788
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 36721
Estimated Expiration: ⤷ Start Trial
Patent: 48467
Estimated Expiration: ⤷ Start Trial
Patent: 71411
Estimated Expiration: ⤷ Start Trial
Patent: 800015
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 6928
Estimated Expiration: ⤷ Start Trial
Patent: 3287
Estimated Expiration: ⤷ Start Trial
Patent: 9461
Estimated Expiration: ⤷ Start Trial
Patent: 0203
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 25574
Estimated Expiration: ⤷ Start Trial
Patent: 2010150865
Estimated Expiration: ⤷ Start Trial
Patent: 2017098998
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Patent: 75871
Estimated Expiration: ⤷ Start Trial
Patent: 31607
Estimated Expiration: ⤷ Start Trial
Patent: 447254
Estimated Expiration: ⤷ Start Trial
Patent: 2018008
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 6531
Estimated Expiration: ⤷ Start Trial
Patent: 8164
Patent: PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6-DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N-(METHYLSULFONYL)ACETAMIDE
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 6318
Estimated Expiration: ⤷ Start Trial
Patent: 5595
Patent: COMPOSICION FARMACEUTICA QUE CONTIENE 2-{4-[N-(5,6-DIFENILPIRAZIN-2-IL)-N-ISOPROPILAMINO]BUTILOXI}-N-(METILSULFONIL)ACETAMIDA. (PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6- DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N- (METHYLSULFONYL)ACETAMIDE)
Estimated Expiration: ⤷ Start Trial
Patent: 11013471
Estimated Expiration: ⤷ Start Trial
Patent: 18006343
Patent: COMPOSICION FARMACEUTICA QUE CONTIENE 2-{4-[N-(5,6-DIFENILPIRAZIN- 2-IL)-N-ISOPROPILAMINO]BUTILOXI}-N-(METILSULFONIL)ACETAMIDA. (PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6- DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N- (METHYLSULFONYL)ACETAMIDE.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 637
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 7352
Estimated Expiration: ⤷ Start Trial
Patent: 2784
Patent: Pharmaceutical composition containing 2-{ 4-[n-(5,6- diphenylpyrazin-2-yl)-n-isopropylamino]butyloxy} -n- (methylsulfonyl)acetamide
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 18015
Estimated Expiration: ⤷ Start Trial
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 181072
Patent: COMPOSICION FARMACEUTICA QUE CONTIENE 2-{4-[N-(5,6-DIFENILPIRAZIN-2-IL)-N-ISOPROPILAMINO]BUTILOXI}-N-(METILSULFONIL)ACETAMIDA
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 015502824
Patent: CRYSTALS
Estimated Expiration: ⤷ Start Trial
Patent: 015502825
Patent: CRYSTALS
Estimated Expiration: ⤷ Start Trial
Patent: 018501161
Patent: PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6- DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N- (METHYLSULFONYL)ACETAMIDE
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Patent: 75871
Estimated Expiration: ⤷ Start Trial
Patent: 31607
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Patent: 75871
Estimated Expiration: ⤷ Start Trial
Patent: 31607
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 56206
Patent: КРИСТАЛЛЫ (CRYSTALS)
Estimated Expiration: ⤷ Start Trial
Patent: 35547
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, СОДЕРЖАЩАЯ 2-{ 4-[N-(5,6-ДИФЕНИЛПИРАЗИН-2-ИЛ)-N-ИЗОПРОПИЛАМИНО]БУТИЛОКСИ} -N-(МЕТИЛСУЛЬФОНИЛ)АЦЕТАМИД (PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6-DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N-(METHYLSULPHONYL)ACETAMIDE)
Estimated Expiration: ⤷ Start Trial
Patent: 12102678
Patent: КРИСТАЛЛЫ
Estimated Expiration: ⤷ Start Trial
Patent: 18123304
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, СОДЕРЖАЩАЯ 2-{ 4-[N-(5,6-ДИФЕНИЛПИРАЗИН-2-ИЛ)-N-ИЗОПРОПИЛАМИНО]БУТИЛОКСИ} -N-(МЕТИЛСУЛЬФОНИЛ)АЦЕТАМИД
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01800077
Estimated Expiration: ⤷ Start Trial
Patent: 02000177
Estimated Expiration: ⤷ Start Trial
Saudi Arabia
Patent: 8391686
Patent: {4- [N- (5، 6- داي فينيل بيرازين -2- يل) -N- تركيبة صيدلانية تحتوي على 2- -N- أيزوبروبيل أمينو] بيوتيل أوكسي} (ميثيل سيلفونيل) أسيتاميد (Pharmaceutical Composition Containing 2-{4-[N-(5,6-Diphenylpyrazin-2-YL)-N-Isopropylamino]Butyloxy}-N-(Methylsulfonyl)Acetamide)
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 791
Patent: ČVRSTI FARMACEUTSKI PREPARAT KOJI SADRŽI 2 {4 [N (5,6 DIFENILPIRAZIN 2 IL) N IZOPROPILAMINO] BUTILOKSI} N (METILSULFONIL)ACETAMID (SOLID PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6-DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N-(METHYLSULFONYL)ACETAMIDE)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201403313W
Patent: CRYSTALS
Estimated Expiration: ⤷ Start Trial
Patent: 201804320Q
Patent: PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6- DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N- (METHYLSULFONYL)ACETAMIDE
Estimated Expiration: ⤷ Start Trial
Patent: 6915
Patent: CRYSTALS
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 47254
Estimated Expiration: ⤷ Start Trial
Patent: 75871
Estimated Expiration: ⤷ Start Trial
Patent: 31607
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1109099
Patent: CRYSTALS
Estimated Expiration: ⤷ Start Trial
Patent: 1804387
Patent: PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6- DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N- (METHYLSULFONYL)ACETAMIDE
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2669213
Estimated Expiration: ⤷ Start Trial
Patent: 2705198
Estimated Expiration: ⤷ Start Trial
Patent: 2759845
Estimated Expiration: ⤷ Start Trial
Patent: 2829415
Estimated Expiration: ⤷ Start Trial
Patent: 120109457
Estimated Expiration: ⤷ Start Trial
Patent: 170024165
Estimated Expiration: ⤷ Start Trial
Patent: 180081141
Estimated Expiration: ⤷ Start Trial
Patent: 240090716
Estimated Expiration: ⤷ Start Trial
Patent: 240090717
Estimated Expiration: ⤷ Start Trial
Patent: 250107951
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 60007
Estimated Expiration: ⤷ Start Trial
Patent: 97124
Estimated Expiration: ⤷ Start Trial
Patent: 30325
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1111352
Patent: Crystal
Estimated Expiration: ⤷ Start Trial
Patent: 1720444
Patent: Pharmaceutical composition containing 2-{4-[n-(5,6-diphenylpyrazin-2-yl)-n-isopropylamino]butyloxy}-n-(methylsulfonyl)acetamide
Estimated Expiration: ⤷ Start Trial
Patent: 31565
Estimated Expiration: ⤷ Start Trial
Patent: 50143
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 8849
Patent: КРИСТАЛІЧНА ФОРМА 2-{4-[N-(5,6-ДИФЕНІЛПІРАЗИН-2-ІЛ)-N-ІЗОПРОПІЛАМІНО]БУТИЛОКСИ}-N-(МЕТИЛСУЛЬФОНІЛ)АЦЕТАМІДУ
Estimated Expiration: ⤷ Start Trial
Patent: 4002
Patent: ТВЕРДИЙ ПРЕПАРАТ, ЩО МІСТИТЬ 2-{4-[N-(5,6- ДИФЕНІЛПІРАЗИН-2-ІЛ)-N-ІЗОПРОПІЛАМІНО]БУТИЛОКСИ}-N-(МЕТИЛСУЛЬФОНІЛ)АЦЕТАМІД (PHARMACEUTICAL COMPOSITION CONTAINING 2-{4-[N-(5,6- DIPHENYLPYRAZIN-2-YL)-N-ISOPROPYLAMINO]BUTYLOXY}-N- (METHYLSULFONYL)ACETAMIDE)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering UPTRAVI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 102099026 | ⤷ Start Trial | |
| European Patent Office | 2447254 | ⤷ Start Trial | |
| Brazil | PI1015936 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for UPTRAVI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1400518 | CA 2016 00048 | Denmark | ⤷ Start Trial | PRODUCT NAME: SELEXIPAG ELLER ET SALT DERAF; REG. NO/DATE: EU/1/15/1083 20160519 |
| 1400518 | 44/2016 | Austria | ⤷ Start Trial | PRODUCT NAME: SELEXIPAG ODER EIN SALZ DAVON; REGISTRATION NO/DATE: EU/1/15/1083 (MITTEILUNG) 20160519 |
| 1400518 | 93266 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: SELEXIPAG OU UN SEL DE CELUI-CI; FIRST REGISTRATION DATE: 20160519 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for UPTRAVI
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.